Disequilibrium
A balloon that does not burn
By Cole Entress
Using discrepant events to confront misconceptions
Water is the most biologically and geologically important liquid on Earth. It is the prototypical liquid to which children (and adults) compare all others (Kind 2004). Yet, through its extensive hydrogen bonding, H2O possesses properties that make it anything but typical. Among these distinctive properties is a high specific heat capacity—that is, it takes a great deal of energy to raise the temperature of a gram of water by only 1°C (1.8°F).
Students are, of course, familiar with heating water to a boil. However, they may not recognize that much more energy is required to raise water to the boiling point (100°C, 212°F) than would be required for an equal mass of nearly any other common liquid (e.g., oil, alcohols) or, for that matter, most solids (e.g., aluminum, concrete, brick, or rubber). Additionally, the temperature of liquid water will not exceed the boiling point. Heat added to boiling water will begin converting it to steam, which can then be heated to temperatures greater than 100°C (212°F).
This month’s Disequilibrium column explores how to help students better appreciate the “heat sink” properties of water—a concept that supports MS-PS1-4 (see NGSS Connections)—using some surprising demonstrations involving fire and water.
Engage
Prepare two latex balloons, as seen in Figure 1. One balloon will be inflated normally. Add 30 ml (1 oz.) to 50 ml (1.7 oz.) of water to the other balloon before inflating it (see Resources for more detailed guidance). Then, light a candle on a clean demonstration table. Tell the class you are going to lower each balloon (attached to the end of a meterstick) to the flame. Ask students to guess how long it will take each balloon to pop. Generally, students will correctly surmise that the air-filled balloon will pop nearly instantly, but will not expect what happens with the second balloon, which will stubbornly refuse to pop, as long as the heat from the flame is focused on the portion of the balloon containing the water.
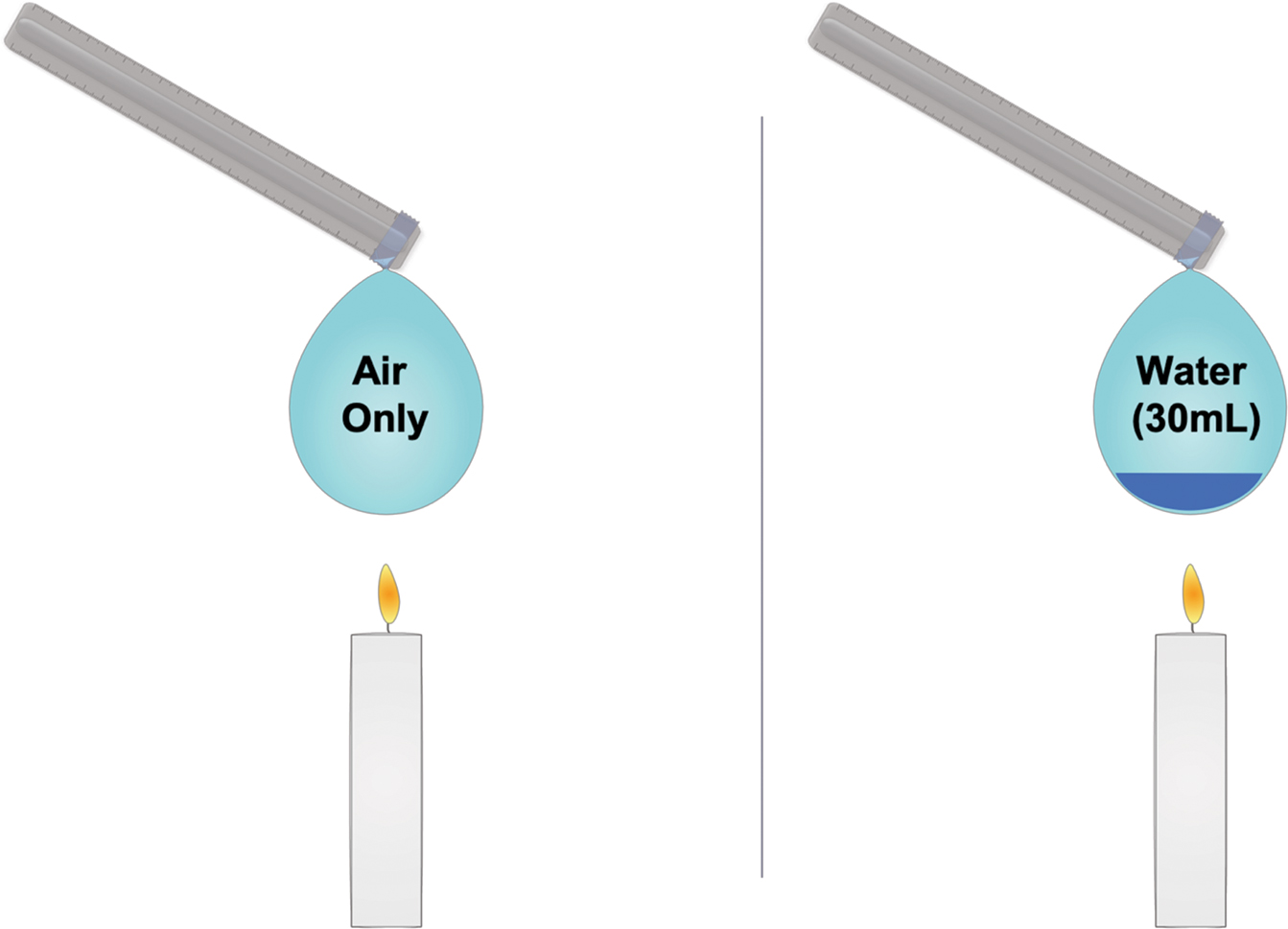
Have a student time the trial with a stopwatch, setting the balloon aside after about 45–60 seconds. Direct pairs of students to try and explain why the second balloon did not pop. Push students, as appropriate, to discuss energy transfer and the particulate nature of matter. If these ideas have not yet been taught, the American Chemical Society has some great ideas for middle grade–appropriate lessons (see Resources).
Explore
Arrange two containers underneath an incandescent light (higher wattage is better) containing equal volumes of sand and water, as shown in Figure 2 (see Resources from Physics Quest for more information). Containers that are wide and shallow are best (8 oz. deli containers or cut-off Styrofoam cups both work well). Place a thermometer in each container so that it is sensing the temperature underneath the surface, and tell students you will turn on the light for 10 minutes, recording the temperature of the sand and the water each minute. Before you begin, ask students which substance they think will heat more quickly, and which will cool more quickly, and why. Then, place the light a few inches from the containers and have students report the temperatures every minute for 10 minutes. Turn off the light and have students report the temperature every minute for another five to 10 minutes. Students will notice that the sand both heats up and cools down more rapidly than the water.

Management notes
If you are worried about classroom management here, there are three possibilities. First, have student groups do the lab, instead of doing it as a class. Second, ask students to create line graphs of the data in “real time,” on whiteboards, chart paper, sheets of graph paper, or with Excel. Finally, to minimize down time, have students record temperature data more frequently—every 30 seconds, for instance.
Explain
Ask students to identify the patterns they saw. Then, introduce and define the term specific heat capacity. When the same amount of energy is added to two substances, the substance with the higher specific heat capacity will have less of an increase in temperature. A higher specific heat capacity also means that a substance must lose more energy in order to decrease in temperature. Specific heat capacity is analogous to inertia in many ways. They are both ways of describing a natural resistance to a physical change.
Ask students to connect this notion to the balloon demonstration at the beginning of the lesson. Push students to identify that the water in the balloon was able to absorb energy from the flame, preventing the latex surface of the balloon from reaching its ignition temperature (incidentally, latex will ignite at about 120°C, 248°F). Also note that water not only has a higher specific heat capacity than the air in the first balloon, but it is also a better thermal conductor (that is, energy is transferred into the water from the latex surface of the balloon far more efficiently than it can be transferred into the air in a regular balloon).
Elaborate
Ask students whether they think that water can be boiled inside a paper cup put directly above a flame. Ask them to justify their predictions, and then show a video (see Resources) demonstrating how a paper cup can be put directly over a Bunsen to boil the water. If your school has adequate facilities and safety equipment, you could do the demonstration yourself.
Ask students to identify the boiling point of water (100°C, 212°F). Tell students that while boiling, water remains at roughly 100°C (212°F)—but the boiling point decreases a bit with altitude, so teachers in Denver beware! Boiling water will remain at this temperature until all the water is converted to steam—it cannot get hotter! (You may want to have a demonstration of this handy, as students may not take your word for it.) Finally, project the cover of the book Farenheit 451, and see if any students know anything about it. Let students know the title of the book is a reference to the temperature at which the paper in books ignites (about 233°C, 451°F). Ask students to compare this temperature to that of boiling water. How might this explain why the paper cup does not ignite? And how else might this be put to use in terms of regulating the temperature of things?
Evaluate
Finally, ask students to research a use of water’s high specific heat in cooking, engineering, industry, medicine, or another field of their choice. For homework, have students write a paragraph explaining how the high specific heat of water—and its thermal conductivity—allow the process to function more efficiently.
Conclusion
Nearly all students will have had experience with water at different temperatures, but few will have been asked to synthesize those experiences into a coherent model. This lesson, though it does not dwell on a molecular-level explanation of water’s specific heat (again, see the American Chemical Society’s website here for lesson ideas), may help students understand water’s high specific heat capacity, and why it matters. In turn, this will help students to make better sense of phenomena such as phase changes and thermal energy transfer.


