FOCUS ON PHYSICS
Emphasizing Physics Concepts With Cartoons, Part II
The Science Teacher—January 2020 (Volume 87, Issue 5)
By PAUL G. HEWITT
Focus on Physics last month featured cartoons that illustrate some of the central concepts in mechanics. We continue with physics beyond mechanics, introducing additional cartoons that I’ve created for future editions of my textbooks. They are first shown here.
Inverse-square laws
One of the nice things about physics is the similarity of its laws. It is actually a small step from understanding the macroscopic gravitational forces of interacting objects to understanding the microscopic electrical forces between charged particles. Both Newton’s law of gravity and Coulomb’s law (Figure 1) relates to forces that follow the inverse-square law.

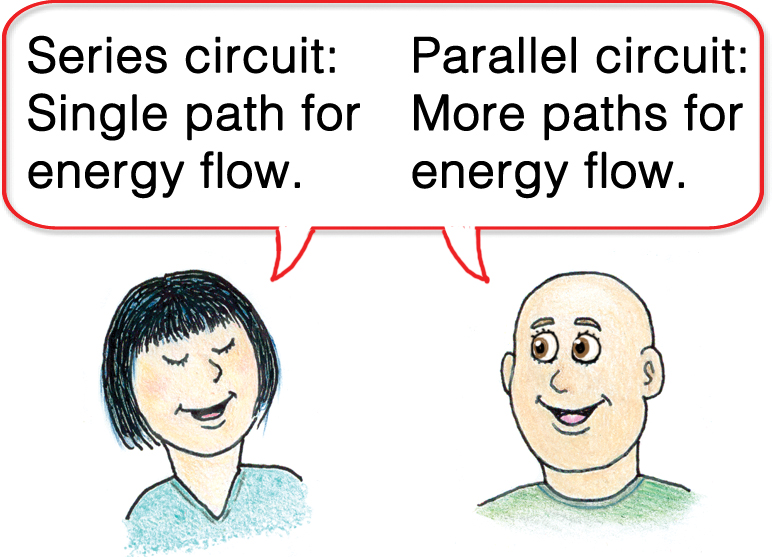
Electric current
A flow of electric charge constitutes the current in electric circuits. When the path of the circuit consists of a single route, we have a series circuit. All devices along a series circuit share the same current. When the circuit has alternate paths, we have a parallel circuit. Energy-consuming devices in an electric circuit can be fed by series or parallel paths (Figure 2).
Ohm’s law
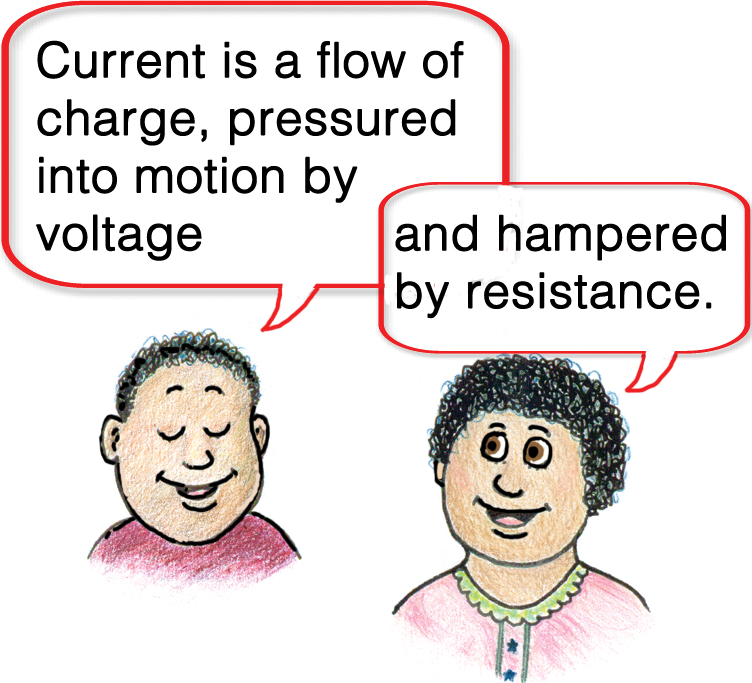
The rule that connects current, voltage, and resistance in a circuit was discovered early in the 19th century by German scientist and teacher Georg Ohm (Figure 3).

When a voltage is applied between two points along a wire or other conductor, the amount of current made to flow is, first, directly proportional to the applied voltage. The greater the voltage, the greater the current. Secondly, the amount of current depends on the electrical resistance—in inverse proportion (Figure 4). So the current in the coils in your toaster depends on both the voltage applied and the resistance of the coils.
Mascot Sneezlee relates all this to what birds do best—sing. In this case in praise of Georg Simon Ohm, for whom the unit of electrical resistance is named.

Electric field
In the space surrounding every electron or proton is what we call an electric field. Just as a gravitational field surrounds an object with mass, an electric field surrounds electric charge. Energy is propelled in electric circuits by the electric field. Although the drift speed of electrons as they move in a current is very slow, the packet of energy carried by the electric field travels at close to the speed of light (Figure 6). Consider a circuit powered by a battery: When the switch in the circuit is activated, the chemical energy stored in the battery converts to electrical energy and sends the electric field instantaneously through all parts of the circuit. The electric signal travels at nearly the speed of light.

Magnetic field
A close cousin to the electric field is the magnetic field. Changes in one field can generate (induce) the other. Oscillations of an electric field, for example, induce a magnetic field that has an identical frequency of oscillation. Oscillating electric and magnetic fields can couple to produce energy-carrying electromagnetic waves.
In about 1860 James Clerk Maxwell calculated the speed of electromagnetic waves to be the same speed as what was already known to be the speed of light. Maxwell identified the nature of light! Light turns out to be a tiny part of the electromagnetic spectrum, ranging from radio waves to gamma rays, all propagating through space at the speed of light, c.
Why this special speed c? If electromagnetic waves travelled faster, energy would be gained. If they travelled slower, energy would be lost. Both faster and slower speeds of electromagnetic waves violate the law of conservation of energy (Figure 7).

Electromagnetic waves
Vibrating electric charges are the fundamental source of electromagnetic waves. All materials are composed of atoms that contain vibrating electrons—all materials radiate electromagnetic waves. The average frequency of the radiated waves is proportional to the absolute temperature of the material emitting them—another central rule of nature (Figure 8).

The relationship f ~T tells us that every body continuously radiates waves of energy with an average frequency proportional to the absolute temperature of the radiating body. Radiation continues without letup! Then why doesn’t the temperature of the radiating body decrease as it steadily gives up its energy? It doesn’t because at the same time it is also absorbing radiation. It both radiates and absorbs simultaneously (Figure 9)!

We tend to think of space as being empty, when in fact space is filled with electromagnetic waves and subatomic particles everywhere (Figure 10).

Electromagnetic radiation and energy
Just as macroscopically the average frequency of radiation is proportional to the temperature of the emitter, also microscopically, frequency determines the energy carried by that radiation (Figure 11).

This central law of physics tells us that the energy carried by a unit of electromagnetic radiation—a photon—is equal to the radiation frequency multiplied by Planck’s constant h. And Manny Mouse also reminds us that emitted energy relates to “quantum jumps” in the atom (Figure 12).

You may want to encourage your students to investigate the energy associated with the electromagnetic waves of their smartphones. High-frequency waves mean high energy per photon; low-frequency waves mean low energy per photon (Figure 13).

Light and color
A particularly delightful part of a physics course is the study of light and color. Lillian illustrates the wonder of addition of red-green-blue (RGB) primary colors (Figure 14).

Cyan, yellow, and magenta (CYM), the subtractive primaries, are seen in thin films (Figure 15).

Nellie Newton makes a surprising point about the color of blue jay feathers in Figure 16.

In Figure 17, Gentleman Gus shares his opinion as to whether knowing the rules of nature adds to its wonder—or subtracts.

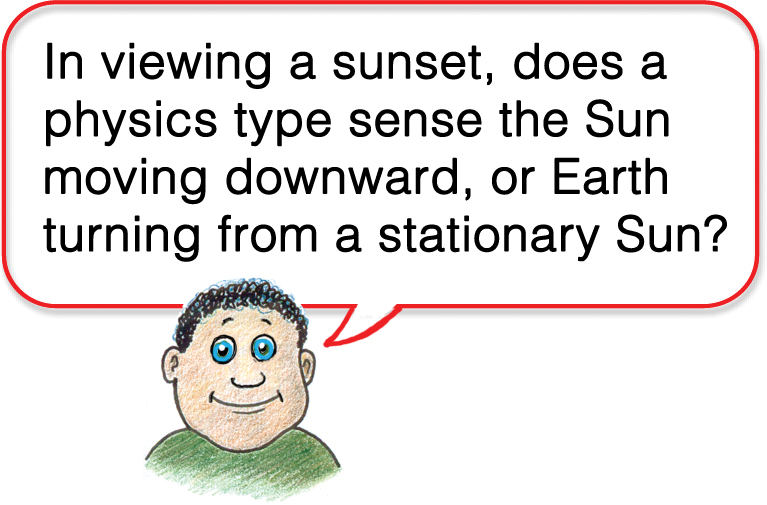
We all know that Earth rotates daily about its polar axis while orbiting a “stationary” Sun. When a point on our ever-twirling planet rotates toward the Sun we have sunrises. When rotation is in a direction away from the Sun, we have sunsets. Phil Physiker ponders this in Figure 18.
E = mc2
We conclude with the most widely known—yet most widely misunderstood—equation of physics. It can be seen as delightful or horrifying, depending on its application (Figure 19). Although the public first learned of this central equation of nature via nuclear bombs, the wider view is that energy and mass are two sides of the same coin. The relationship between energy and mass encompasses all energy transfers in chemistry and in biology, underlying all the energies associated with life itself.

All the equations here are central to physics, and to the rules of nature. My strongly felt view is that physics taught as the study of nature’s rules should be in the educational mainstream for all students—those who will go on to study science, as well as those moving on different educational paths. Nature is central to all.


