Feature
Patterns, Puzzles, and the Periodic Table
Teaching the periodic table is a central part of chemistry. This might involve memorizing the symbols of the elements, learning about the properties of the families, or understanding its general organization. This year, The International Year of the Periodic Table, marks its 150th anniversary, and represents a good opportunity to highlight its history.
The underlying patterns described by periodic law, such that the table is organized horizontally by the number of protons and vertically by similar chemical properties in addition to the repeating patterns associated with those electrons found in the outermost shell of an atom correlate with the Next Generation Science Standards (NGSS) Disciplinary Core Idea (DCI) Structure and Properties of Matter (PS1.A) (NGSS Lead States 2013). Furthermore, observing patterns and the relationships underlying them is one of the key crosscutting concepts found in the NGSS (see NGSS connection box, page 35).
The following lesson requires about two traditional class periods or one block period and follows the 5E Instructional Model designed by the Biological Sciences Curriculum Study (Bybee 2014) with the added Elicit stage proposed by Eisenkraft (2003). Students first emulate Dmitri Mendeleev’s own experience with organizing the elements through a puzzle activity, using patterns to make predictions. Then after reading a non-fiction text about his contributions (Bryson 2008), students gain a foundational understanding of the patterns within the periodic table before spending time exploring the properties of the families.
Elicit
Start class by asking students about their experiences with puzzles, if they like puzzles, and what kinds of puzzles they like. Students spend time sharing about their experiences with puzzles and connecting them to their own interests. Explain that today they will be working on some different kinds of puzzles.
Students should have experience with the ideas of Carol Dweck and understand the difference between having a growth mindset and fixed mindset, and what it means to have a mastery response or a helpless response (Dweck 2006; Mindset Works 2016). This is important because many students find science challenging, and having these conversations in the beginning starts to create a culture that welcomes mistakes as part of the learning process. Students are informed that the puzzle will be challenging, and they need to draw on their growth mindset. Reinforce that they absolutely can complete the puzzle and figure out the pattern. In order to do so they will have to persevere.
Before starting the puzzle, the teacher asks the essential question “Where have you seen patterns in your life?” In groups of three to four students they will respond to this question using the Chalk Talk strategy (Ritchart, Church, and Morrison 2011). Students gather around a whiteboard or large poster paper with the essential question written in the middle. Each student has a different colored marker. Set the timer for two minutes and during that time students silently write, draw, respond, pose new questions, and react to each other’s responses. Then after the silent dialogue, groups are allowed to talk and discuss what everyone wrote or drew. Students can also do a gallery walk around the room to look at other groups’ ideas. In a final discussion the teacher verbally synthesizes their thoughts and looks for themes across the different groups.
Engage and Explore
Students can stay in their Chalk Talk groups or they can be shuffled into new groups. When thinking about the groups, put students together who are going to encourage each other when they struggle.
The puzzle, Understanding the Periodic Table, is a 24-piece set by American Educational, which can be found through science supply stores and online retail distributors at $74.99 (Figure 1). Science teachers could also create their own puzzle if science supply funds are an issue.

Brief the students on the puzzle. The pieces all have a color, whole number, and decimal number with varying numbers of stars, holes, and/or notches on the side. Students have two goals:
- Determine a way to arrange the pieces of the puzzle demonstrating a pattern for each characteristic (color, number, decimal, stars, holes, notches). Before passing out the puzzles, make sure to remove one or two pieces.
- Predict the characteristics of the missing pieces based on evidence from the pattern they developed.
With the right amount of time, every group can figure out the pattern. Some groups will need more time, some groups will need less time. Let students know ahead of time that there are clues should they get really stuck. After 10 minutes, if they do not have at least a pattern for two of the characteristics, students should ask for a hint.
The first hint is related to color. Students are asked about the most common color pattern and most respond with the rainbow pattern or ROY G BIV. Encourage them to arrange the pieces into the rainbow order. Once in the rainbow configuration, students start to see a pattern for the shapes (holes, stars, notches) and whole numbers. When students notice a discrepancy in the pattern for the shapes, guide them to think about their missing piece. A 6” × 4” arrangement (see Figure 1) allows students to most easily see the pattern (this can be another hint), but other dimensions will work.
Finding a pattern for the decimal numbers happens at the very end for most groups. Having four-function calculators on hand is helpful, as some groups want to do quick calculations to look for a mathematical pattern.
Once students have a pattern for the puzzle, they start to work out the missing pieces. When groups are ready, they describe the characteristics of the missing pieces, justifying what the pieces should look like based on the pattern they have discovered. If they are correct in their prediction, give them the missing pieces and remind them to keep the details of the pattern a secret from other groups still working.
Encourage successful groups to help other groups without giving the answer away. If they are incorrect and there is still time, let them continue to work. If time is running out and they have all of the characteristics except the decimal number, then give them the piece to discuss with their group about where they made the error.
Explain
Bring the students back into one large group, explaining that Dmitri Mendeleev (also spelled Mendeleyev), a Russian chemist, had a parallel experience. With only 63 elements having been discovered in 1869, he was also trying to solve an incomplete puzzle. Inspired by the game Solitaire, he put the names, weights, and properties of the elements on cards, moving them around and re-arranging them until a pattern made sense.
What made Mendeleev particularly remarkable was his confidence in the pattern he observed for the arrangement of the elements, which led him to leave gaps in the periodic table for elements not yet discovered, and to make stalwart predictions of the properties of those undiscovered elements. This is similar to having the students moving their puzzle pieces around, looking for a pattern, justifying the pattern based on evidence, and predicting the characteristics of the missing pieces.
Because of the varying reading levels of students across chemistry classes, I have included several resources for articles about Mendeleev and the periodic table (see References) outlining Mendeleev’s struggle, his process for organizing the elements, and his discovery of periodic law. Teachers can choose one article or a few articles and differentiate based on students’ reading levels. Students can read the article individually or out loud to each other in small groups.
After reading the articles, students work in their puzzle groups, answering a series of questions (see Figure 2) connecting their experience with the puzzle to that of Mendeleev’s with the elements. The goal is for students to recognize the natural emergence of patterns in science as well as the struggle scientists go through when developing explanations. Pre-select a speaker from each group to encourage a broad range of responses during the large group discussion. Encourage students to share about their own struggles, persistence, and pattern development.
After students have learned more about Mendeleev and his contributions, they move on to learn more about the modern periodic table and the specific element families. Show a picture of a family or ask students to share pictures of their own families. Pose the question, “What do families have in common?”
In small groups, have students discuss their ideas, then ask someone who has not shared with the large group yet to share. Students usually mention how families share similar body features, behaviors, mannerisms, hobbies, interests, views, and beliefs. Physical traits are the most prominent responses; this allows the teacher to make the connection between the physical traits that people in families share to the physical and chemical properties that elements in families share.
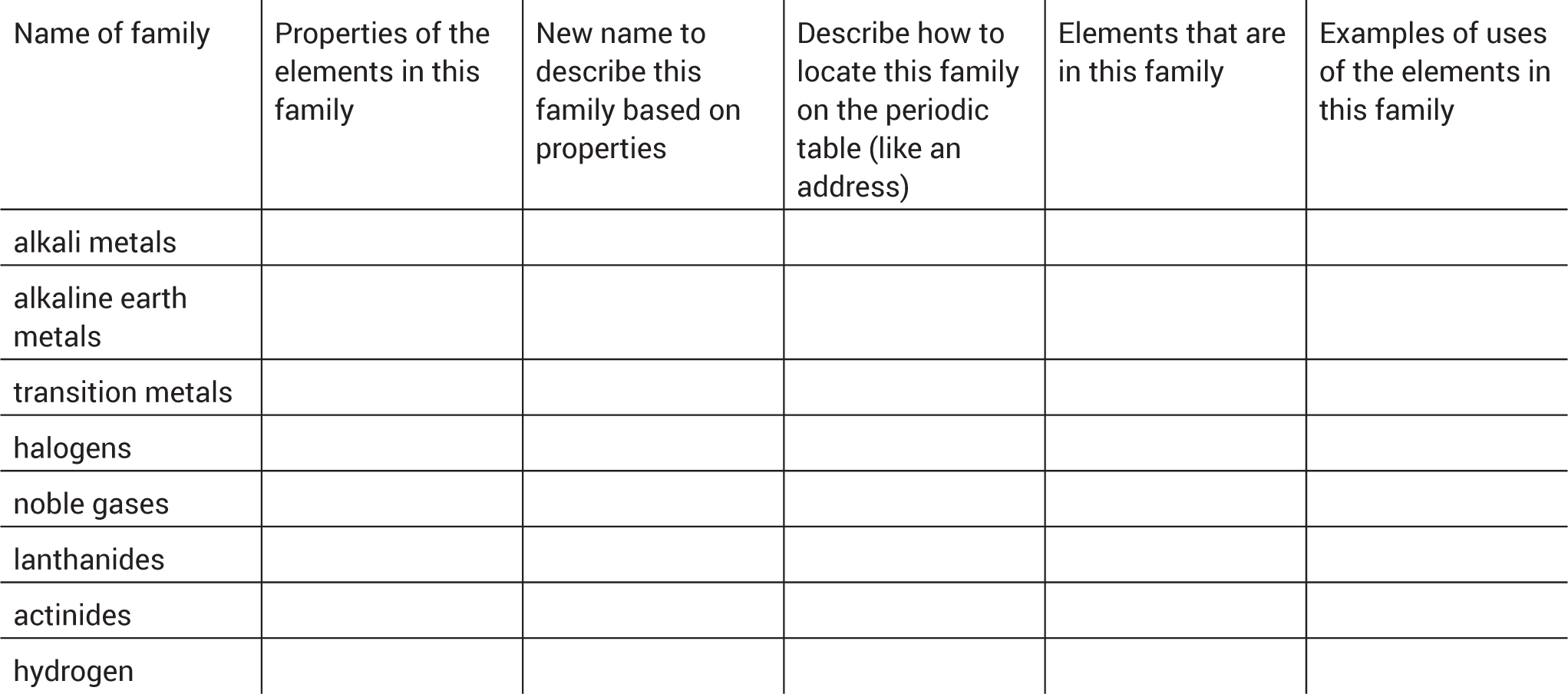
Through a jigsaw activity involving a large felt periodic table (see Figure 3), students learn more about the families and their properties. Put students into seven groups and assign each group a different family from the periodic table: alkali metals, alkaline Earth metals, transition metals, halogens, noble gases, lanthanides, and actinides. Hydrogen is not in a specific family, so the teacher can model the activity with hydrogen or assign it as an eighth group.

The students’ task is to become experts on their assigned family by researching its chemical and physical properties, uses, location on the periodic table, and the elements in that family (see Figure 4). Each group prepares notecards with the symbols of the elements to place on the giant-size periodic table. Use index cards that come in assorted colors and assign one color per family, so students can clearly see the location on the giant-size periodic table.

The class then constructs the periodic table family by family by placing their prepared notecards on the giant-size periodic table using double-sided tape or Velcro (see Figure 4, p. 33). Groups present their findings and take notes during the presentations (see Figure 5 for a sample graphic organizer). After everyone has presented, give the students time to stand back and reflect on the periodic table they have constructed. Ask them what new patterns they observe and respond to any follow-up questions.
Chemistry Crosscutting Concepts



