feature
Teaching Green Chemistry Through Student-Generated Open Educational Resources
Journal of College Science Teaching—March/April 2023 (Volume 52, Issue 4)
By Krystal Grieger and Alexey Leontyev
This article describes the implementation of a student-generated open educational resource project into a majors’ organic chemistry laboratory. This scaffolded semester-long project was organized into six phases and designed to promote the goals of teaching students about both green chemistry and real-life applications of the reactions covered in the course while simultaneously creating open educational resources for future students. A project-based learning approach was utilized to promote student engagement, especially during remote instruction. To further promote engagement, multiple opportunities for peer feedback were provided using web-based software that supports anonymous feedback. The effect of the project on cognitive learning gains and student perceptions was assessed and is reported. Overall, positive learning gains on green chemistry knowledge and skills were observed, and most students viewed the project favorably.
Integrating green and sustainable chemistry in education is essential because it promotes systems thinking and supports meaningful learning (Mahaffy et al., 2019; Zuin et al., 2021). Green chemistry has been deemed an interdisciplinary science because it requires knowledge and expertise from a variety of disciplines to achieve its goal of a sustainable future (Chen et al., 2020). However, a key barrier to its implementation in college curricula is the lack of instructional resources, particularly textbooks, that integrate green chemistry with course content (Andraos & Dicks, 2012; Aubrecht et al., 2019; Grieger, Hill, & Leontyev, 2022; Johnson et al., 2020).
This barrier can be surmounted through open educational resources (OERs) that allow for the customization of the curriculum materials. The use of OERs has been shown to be as effective as traditional textbooks, and courses with OERs were found to have significantly lower withdrawal rates than those that use a commercial textbook (Clinton & Khan, 2019). One platform for the creation and dissemination of OERs is LibreTexts (n.d.), the successor of ChemWiki. LibreTexts allows instructors to create a course text using either their own or previously created OER materials and customize it to fit their needs. LibreTexts includes materials for a variety of science, technology, engineering, and mathematics (STEM) courses, including biology, chemistry, engineering, geosciences, mathematics, medicine, physics, and statistics.
Although the ability to edit and customize course material in OERs is beneficial, one limitation is the time required to create or modify the material. Therefore, one method for promoting the development of green chemistry material in OERs while actively teaching the content is to involve our students in the creation of the material. Creating student-generated instructional materials is a form of project-based learning that provides a variety of benefits for students, including increased motivation due to real applications of their work, a deeper understanding of the content matter, and improved communication and explanatory skills (Coppola & Pontrello, 2020). Recently, positive student feedback on the integration of student-generated instructional materials has been reported within laboratory courses (Grieger & Leontyev, 2020, 2021). In addition to the benefits offered by student-generated instructional materials, the use of writing-to-learn activities has been shown to promote conceptual learning, critical thinking, and the development of disciplinary practices (Finkenstaedt-Quinn et al., 2021). Furthermore, writing-to-learn activities encourage students to “translate” the material into understandable terms for the desired audience, rather than solely regurgitating the instructor’s terminology, as the audience will likely be unfamiliar with the technical jargon (McDermott & Kuhn, 2011). Due to these combined benefits, we developed a semester-long project that integrated discipline-based writing in which students developed an OER describing one of the reactions taught in Organic Chemistry II along with its application in a reaction from literature that used a bio-derived molecule.
Description of the OER activity
This project was conducted at a medium-size midwestern university in the spring semester of 2021, with the approval of the Institutional Review Board (Protocol # SM20271). The students (n = 20) were enrolled in a chemistry majors’ section of Organic Chemistry II lab that met twice a week for 3 hours each time. Due to the ongoing COVID-19 pandemic, the class was split in half, with each half attending in person to conduct experiments and attending remotely in alternate weeks. During the remote weeks, students completed virtual simulations and the OER project. This project was worth approximately 16% of the total score. Other assignments in the course included prelab quizzes, lab reports, problem sets, worksheets, and virtual simulations.
The overall goal for incorporating the OER project was for students to make connections between the organic reactions they were learning about with both green chemistry and real-world examples of the reactions. Therefore, students were asked to research and report on the purpose, mechanism, and history of their selected reaction. Students were then instructed to locate and summarize a literature example of the reaction and evaluate its adherence to green chemistry principles and its effect on the planetary boundaries (Rockström et al., 2009). The planetary boundaries consist of nine boundaries that outline a safe operating space for humanity with consideration for Earth’s systems. Crossing the threshold of the outlined boundaries increases the risk of global environmental change (Rockström et al., 2009). This activity was designed to promote the student learning objectives (SLOs) outlined in Figure 1.
Student learning objectives (SLOs) for the activity.
- Students will evaluate real-life examples of reactions from Organic Chemistry II using the green chemistry principles and planetary boundaries.
- Students will calculate the process mass intensity and atom economy of reactions.
- Students will make connections between concepts in organic chemistry, green chemistry, and real-life practices by creating their open educational resources (OERs) and reviewing their peers’ OERs.
- Students will develop information literacy by locating, evaluating, and using literature in the creation of their OER.
- Students will develop professional communication skills by creating an OER designed for organic chemistry students and by participating in scientific discourse with their peers.
Timeline of the activity
This scaffolded project was organized into six phases (illustrated in Figure 2) to help guide students through its completion. The instructions and feedback guidelines for each phase are provided in the online appendix. Peer feedback was integrated into the first five phases using the platform FeedbackFruits Peer Review (SLO5). Additionally, after making revisions based on the peer reviews, students submitted the first four phases for grading and feedback from the instructor so they could make final revisions before completing the project.
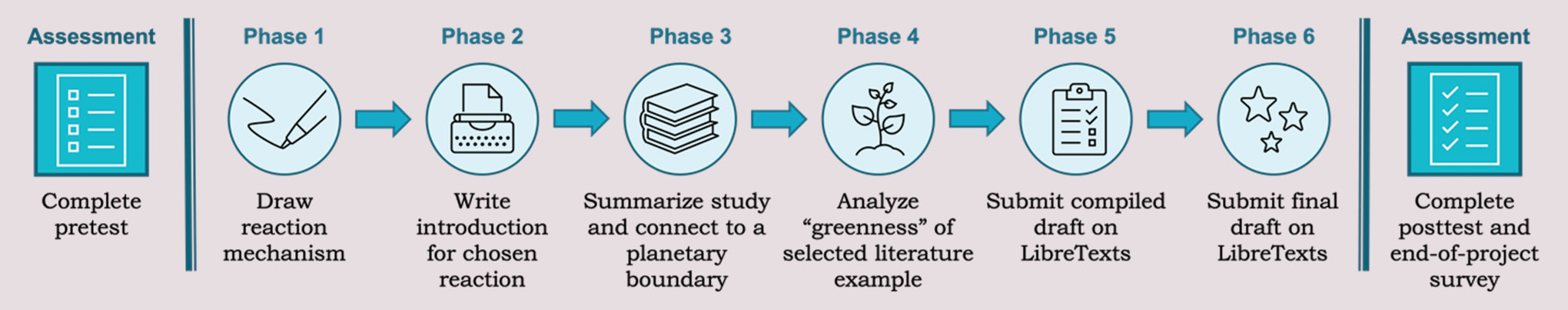
Phases of the OER project.
To assess knowledge and skills gained through completing this project, a pre- and posttest design was incorporated in which students’ knowledge of green chemistry was evaluated prior to and upon completion of the project. To assess student perceptions of the project upon completion, we also had students complete an anonymous survey consisting of a series of open-ended prompts and Likert-style questions.
In Phase I, students each chose a reaction from a list of 39 key reactions that the instructor selected from a second-semester organic chemistry textbook and posted their choice using the discussion board in our classroom management system. Posting of their selection was incorporated to ensure that each student had a unique reaction because students could not select a previously chosen reaction. Students then used free chemical drawing software to draw the reaction mechanism for their chosen reaction.
In Phase II, students drew the general reaction scheme for their selected reaction, identified the interconversion of functional groups that the reaction facilitates, identified functional groups that would be sensitive to the reaction conditions and would require protecting groups, and provided a description of the reaction mechanism.
This led to Phase III, which required students to delve deeper and complete literature searches using SciFinder (SLO4). In this phase, students identified the first report of the reaction and modifications that had been made to it. Next, they found a primary literature source in which the selected reaction was conducted using a bio-based or bio-derived molecule as a reactant, product, or catalyst. Students then drew the selected reaction from the article using free chemical drawing software. Students provided a summary of the article in which they identified the purpose of the study and summarized the findings of the study. Finally, using their understanding of planetary boundaries, students connected the research to a planetary boundary and indicated whether the study would help prevent the boundary’s threshold from being crossed.
In Phase IV, students analyzed their selected reaction from the primary literature article for its adherence to the green chemistry principles and calculated its atom economy and process mass intensity (SLO1 and SLO2). Because some of the literature articles did not include all quantities of reagents that were used, especially for the workup procedure, students were instructed to calculate the process mass intensity using provided quantities but to add a statement that lists the reagents that were not considered.
Next, in Phase V, students updated and copied the information from the previous four phases onto their respective pages on LibreTexts (SLO5). The five pages included (i) information about the reaction, (ii) reaction mechanism, (iii) use of the reaction with a bio-derived molecule, (iv) analysis of the reaction’s adherence to the green chemistry principles, and (v) connection to planetary boundaries. To complete this phase, students were provided with access to the team Sandbox on LibreTexts and editing access for their OER web page. Through completing this phase, students learned about the use of alt text for images and utilized the Mathpix Snipping Tool (Mathpix, n.d.) to convert their equations for atom economy and process mass intensity into Latex for the LibreTexts web pages.
Finally, in Phase VI, students updated their pages based on the peer feedback received and submitted the final copies of their OER pages. A permission form was then signed by students (n = 6, 32%) who wanted their pages to be available online.
Results and discussion
To assess the impact of this activity, a pre- and posttest design was utilized along with an end-of-semester survey to assess students’ perceptions of the project. Within the survey, students were asked to provide a self-assessment of learning gains (SALG). SALG-style questions have been used extensively in research to collect data on student-reported learning outcomes since the format’s development in 1997 (Scholl & Olsen, 2014). Analysis of the SALG responses (as shown in Figure 3) indicated that student knowledge about green and organic chemistry mostly increased and that students had a better understanding of how green chemistry connects to real-life applications (SLO3).
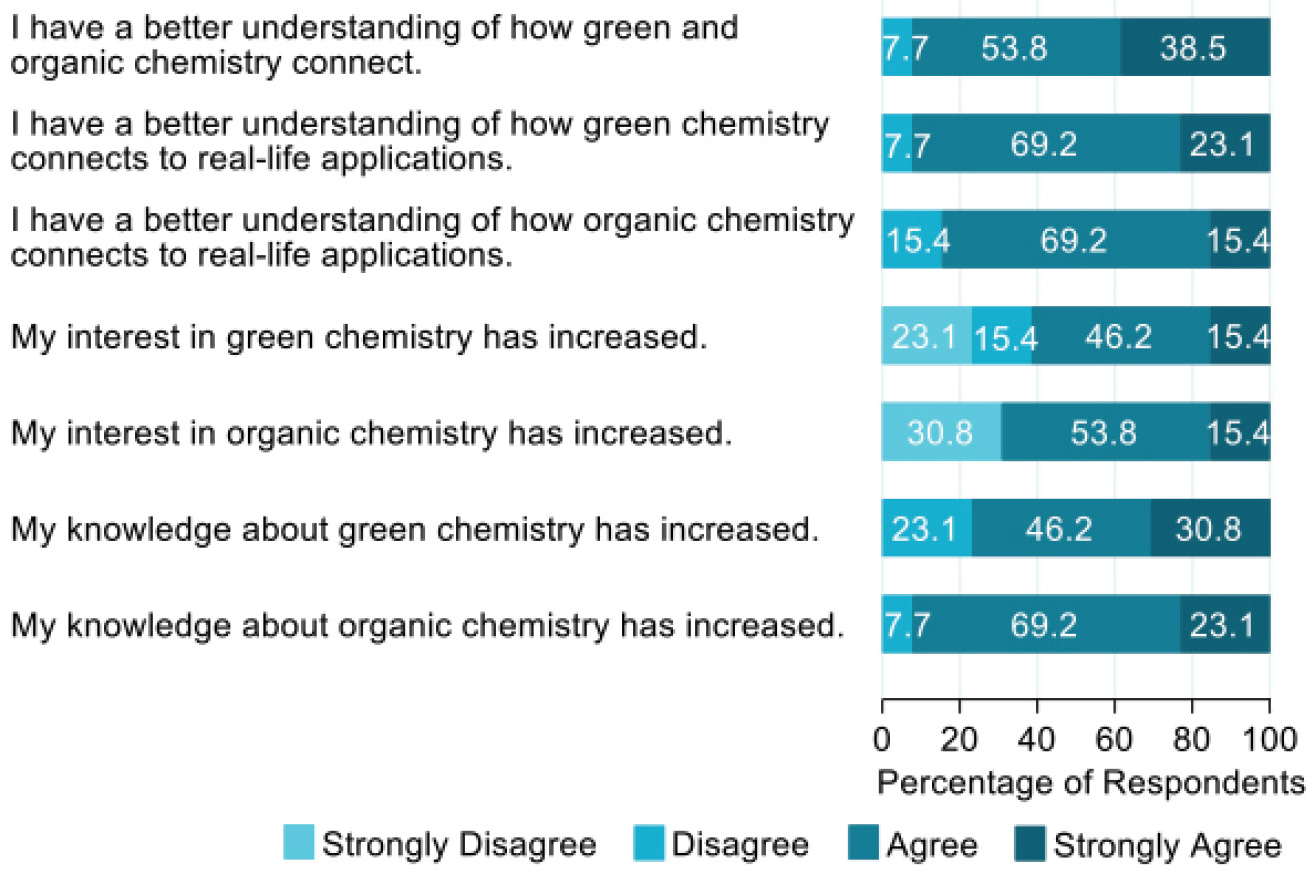
Distribution of student responses for self-assessment of learning gains upon conclusion of the OER activity.
Additionally, students were asked to assess whether they gained disciplinary skills by completing the project. Most students reported that they felt more comfortable communicating chemistry to a general audience and using tools such as chemical drawing software and SciFinder. Full results of the students’ self-assessment of disciplinary skills gained are provided in the online appendix.
Although these results are encouraging, research has shown that self-reported measures are not always reliable indicators of cognitive learning gains (Armstrong et al., 2019). Therefore, we included additional assessment methods to measure the effects of the activity on student learning. First, student knowledge of the green chemistry principles was assessed using an in-house developed Assessment of Student Knowledge of Green Chemistry Principles (ASK-GCP; Grieger, Schiro, & Leontyev, 2022). This assessment contains 24 true-false statements, with one true and one false statement per principle. The average scores for the pre- and posttest are illustrated in Figure 4. The scores from the ASK-GCP pretest (M = 16.9, SD = 0.8) and posttest (M = 19.5, SD = 0.7) were analyzed using a paired t-test to compare the difference in the two population means. The results indicate that completion of the project resulted in a significant increase in knowledge about the green chemistry principles, t(16) = 2.92, p = 0.010. Furthermore, Cohen’s d, which is the standardized mean difference between pre- and posttest, for this analysis (d = 0.708) indicates that, on average, students’ scores increased by more than two thirds of a standard deviation upon completing the activity. Due to the relatively small sample size, to determine the practical significance, we calculated the adjusted effect size, ε² = 0.411, which indicated a strong relationship where approximately 41% of the variability of the posttest score is explained by completing the OER project.
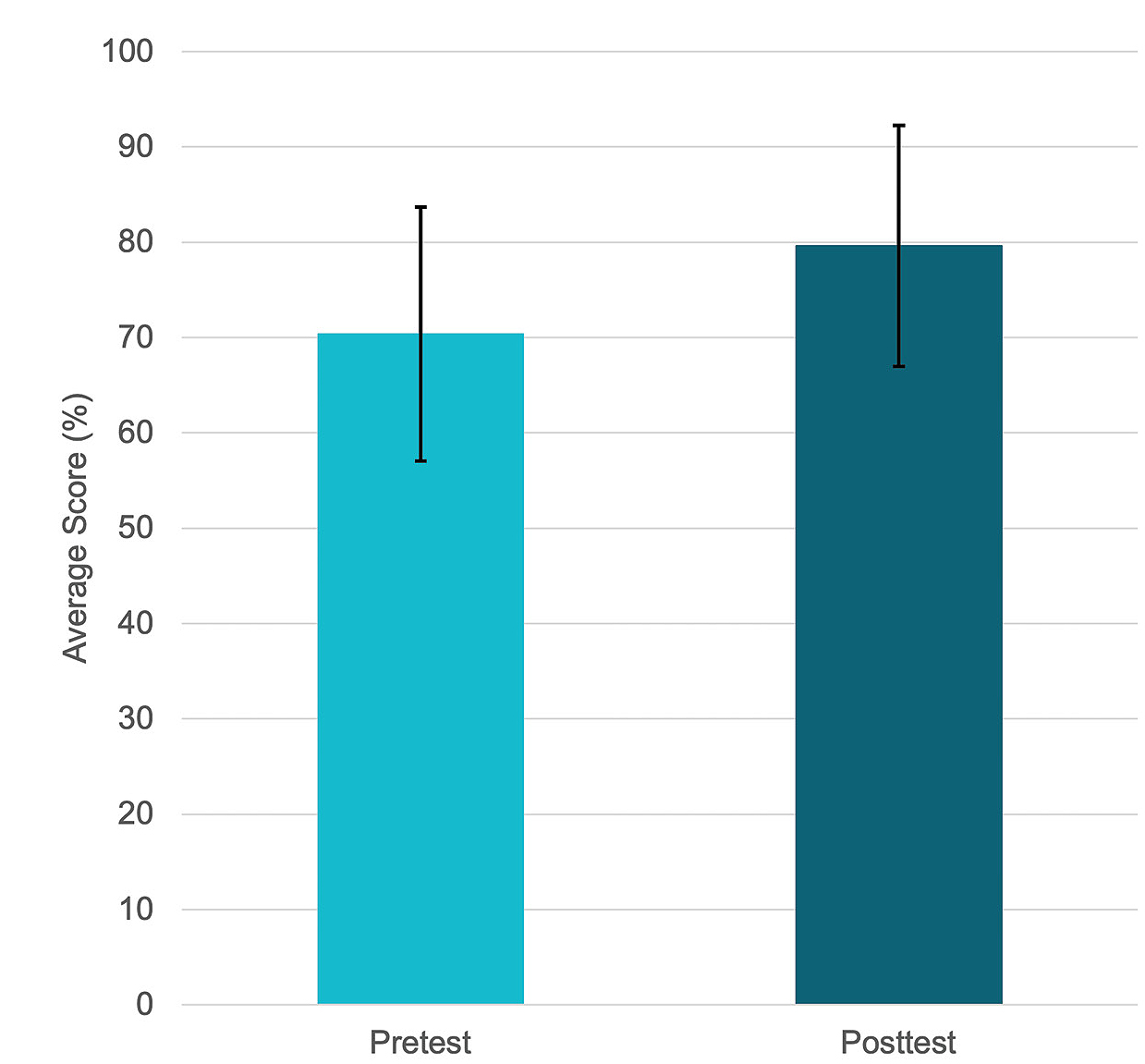
Comparison of pre- and posttest Assessment of Student Knowledge of Green Chemistry Principles scores.
To further assess students’ ability to apply the green chemistry principles, using a prompt by Armstrong et al. (2019), we asked students to identify up to five specific factors that they would consider when deciding which of two reactions yielding the same product was greener. Because most students had been taught about the green chemistry principles and were exposed to similar-style questions in the first semester, students only identified 0.7 more principles on average in the posttest than on the pretest, indicating a ceiling effect for this prompt.
One of the goals of the project (SLO1) was to introduce the concept of planetary boundaries to students. To assess student knowledge about planetary boundaries, we asked them to define the boundaries in the pre- and posttests. In the pretest, 15 students (79%) indicated that their responses were a guess, and none of the responses were fully correct. However, in the posttest, only two students (11%) indicated that their responses were a guess, and 10 (53%) of the students provided a correct definition.
After evaluating the activity’s impact on student learning, we utilized an anonymous survey containing Likert-style and open-ended prompts to assess student perceptions of the project to help us direct future iterations of the project. Overall, when asked if they would like to see this project implemented in future labs, nine students (60%) indicated that they would, five (33%) indicated that they would not, and one (7%) was unsure. Similarly, when asked if they would like to see this project implemented in future lecture courses, 10 students (67%) indicated that they would, whereas five (33%) indicated that they would not.
When asked about beneficial aspects of the project, the most common responses included participating in the peer review process (n = 5), gaining a deeper understanding of reaction mechanisms (n = 3), learning about different reactions (n = 2), learning how to distinguish between primary and secondary sources in literature (n = 2), becoming more familiar with chemical drawing software (n = 2), and having the project completed in phases (n = 2).
To refine the assignment for future iterations, we asked students what aspects of the project they would like changed. The most common responses included rearranging the components of the phases so they are consistent with each book page (n = 3), requiring fewer comments in the feedback for the final phase (n = 3), starting the project earlier in the semester (n = 2), and completing the project in person (n = 2).
Finally, students were asked if they felt what they learned in the project would benefit them in their future careers. Eight students (62%) indicated that it would and two (15%) indicated it might, depending on their future career. Of those indicating that the project would benefit them, the most common reasons included learning how to effectively research a topic (n = 4) and gaining problem-solving skills (n = 2).
Overall, this project resulted in the creation of OERs for eight different reactions from Organic Chemistry II that were publishable as submitted. Among those that would require revisions for publication, the most common errors included errors in analysis of “greenness” of the reaction (n = 9), an incorrect description of the reaction mechanism (n = 4), and an incorrect or vague description of the relation of the research to a planetary boundary (n = 3). With regards to the application of the green chemistry principles, the most common error involved the application of real-time analysis for pollution prevention (n = 6). Responses providing incorrect claims about how this principle applied to the reaction included that the product was analyzed for purity, that the reaction was monitored for completion via thin layer chromatography, and that the reaction must have been monitored due to a longer reaction time. The variety of responses received for each page illustrated a key advantage of having each student report on a different reaction and different literature article because doing so helped ensure that all student responses were their own, as students could not copy answers from their peers or the internet. This is of particular importance because this project has been shown to prevent student cheating, which has been a concern of instructors with the transition to online instruction in particular (Nguyen et al., 2020).
Conclusions
The incorporation of student-generated OERs allows instructors to teach about green and sustainable chemistry while creating resources for our own courses and other courses. The materials prepared through this project can be used to integrate examples of relevant reactions with bio-derived molecules into Organic Chemistry II.
Creating student-generated OERs helps motivate students to gain a more thorough understanding of the topic because they must “explain” it to incoming students in a resource that has a purpose beyond just demonstrating their knowledge to their instructors.
This project was designed to both create an OER and teach our students about green and sustainable chemistry. This project was perceived favorably by most of our students and resulted in positive learning gains for the green chemistry principles and planetary boundaries. Although the sample size was small, this was by design because it allowed for piloting the project within one section to assess its impact. Future work will include scaling up the project’s implementation and integrating student-generated questions on green chemistry (Zurcher et al., 2016). All resources for implementing this project have been provided in the online appendix. It is our hope that by disseminating this model, we can promote adoption of OERs by practitioners of various STEM disciplines while creating a wealth of academically relevant materials for all to use. Through creating the OERs, it is our hope that more instructors incorporate the use of OERs in their classrooms because they lower the cost for students, which subsequently promotes equality.
Acknowledgments
Research presented in this article was supported by the National Science Foundation under NSF EPSCoR Track-1 Cooperative Agreement OIA #1946202. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation. In part, this research was also supported by NDSU’s NDUS ECOR Award FAR0035075.
We wish to thank the students who participated in this project and responded to the survey. We would especially like to thank Delmar Larson and Joshua Halpern for setting up the course page on LibreTexts and providing guidance on the project. We would like to thank Brent Hill for his help with the statistical analysis of the pre- and posttests. Finally, we would like to thank Jose Medina for his help with analyzing the student perceptions of the project.
Krystal Grieger is a doctoral student in the Department of Chemistry and Biochemistry, and Alexey Leontyev (alexey.leontyev@ndsu.edu) is an assistant professor in the Department of Chemistry and Biochemistry and Discipline-Based Education Research PhD Program, both at North Dakota State University in Fargo, North Dakota.
Chemistry Teacher Preparation Teaching Strategies


