Constructive Error Climate: A Classroom Assessment Technique in Science Classes
Although making errors in the learning process is common, it is usually perceived by students as something negative and a potential threat to self-esteem. Such perception often prevents students from considering errors as learning opportunities. By using errors as integral elements of the learning process within the science classrooms, and establishing constructive error climate, students can learn from errors and the learning process is fostered.
Background
As educators, we often assume that our students are learning what we teach. However, the contrary fact is, students may not have learned as much as expected: gaps exist between what was taught and what was learned. It is usually late when we collect the information from students’ homework, quizzes, and exams, as students think of anything they have been tested on as being completed. Consequently, the more effective moments of assessment are before the homework or tests. Classroom assessment techniques (CATs) are generally simple activities aiming at providing early feedback and continual accurate information to assess student learning throughout the semester (Cross & Angelo, 1993). CATs are particularly useful for checking how well students are learning at early stage, and providing information for improvement when learning is unsatisfactory. With proper use of CATs, instructors are able to promote both teaching and learning and help students become more effective and self-directed learners (Steadman, 1998). Even though many CATs have been developed and successfully incorporated into the classrooms (Cross & Angelo, 1993; He, 2019; Cauley & McMillan, 2010), this article focuses on a topic that has rarely been discussed in chemical education, but is beneficial to students learning science: error climate (Steuer, Rosentritt-Brunn, & Dresel, 2013).
Errors are a natural and common by-product in the learning process, and frequently perceived by students as something negative and shameful. If more errors appear, students are more likely to get poor grades in the class. This perception often prevents students from seeing errors as learning opportunities (Wenzel, 2002). Mistakes are the stepping stones to learning, thus errors could help establish accurate mental models and foster the learning process. From the theory of negative knowledge, reflecting on errors can label incorrect parts of an initial mental model as incorrect and reorganize what is right, resulting in construction of a more comprehensive mental model (Gartmeier, Bauer, Gruber, & Heid, 2008). Certainty is gained because correct and incorrect conceptions can be more clearly distinguished.
Students’ reactions to errors, shame, or learning opportunity depend not only on their individual characteristics, but also on the learning environment (Wenzel, 2002), and an appreciative handling of errors in the classroom is advantageous. A constructive error climate uses errors as integral elements of the learning process within the classroom, which can lead to the construction of stable knowledge, the improvement of emotional states, and better student performance (Steuer & Dresel, 2015). Some instructors have realized the value of students’ mistakes and include an assignment of analyzing mistaken statements from students’ test answers (Yerushalmi & Polingher, 2006). Recent educational research suggests that learning through mistakes can potentially foster students’ knowledge, and a constructive error climate in the learning process enhances the quality of learning, leading in turn to stable knowledge and improved achievement (Steuer & Dresel, 2015; Wernecke, Schütte, Schwanewedel, & Harms, 2018).
Implementation
One simple and effective CAT to establish a constructive error climate is proposed; this CAT has been implemented in my chemistry classes for two academic years. The class size is normally under 24 (the maximum capacity of one class in this college), but this CAT could also be incorporated into larger size science classes.
Three chemistry topics “Naming Ionic Compound,” “Writing Ionic Compound Formulas,” and “Writing Ground State Electron Configuration” are used as examples, as shown in Figures 1–3. This technique can be adapted into many other topics in science teaching. Using Figure 1 as a demonstration, after the topic “Naming Ionic Compound” is taught, students work in small groups (usually groups of 2–4), and discuss and discover some errors, as shown in Figure 1a. Students in the same group work constructively to recognize the misconceptions and figure out the reasons behind the errors. The exchange of ideas leads students to deal with the mistakes at the level of “why the mistake is made” and “how to solve the mistake,” rather than just memorizing “what is the correct answer.” When students finish, the instructor may ask students to share their answers between groups. The next slide with correct answers is then displayed, as shown in Figure 1b. The errors are bolded in red, and the correct answers are provided and bolded in green. With this contrast, it is easier for students to compare their group answers with the ones provided by the instructor and get a better understanding of the errors.
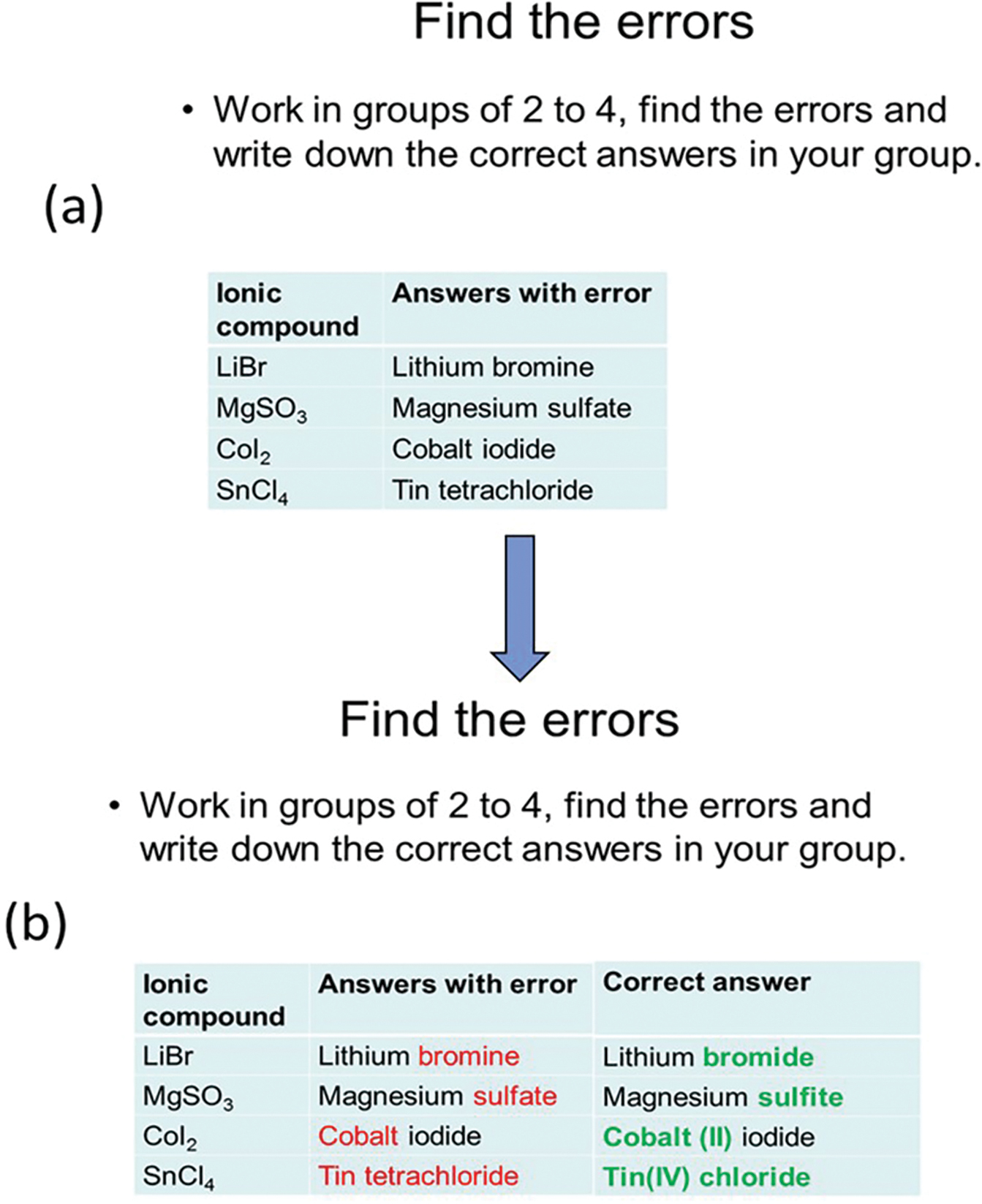
(a) The slide after the topic “Naming Ionic Compound” is taught. Students will work in groups to find and correct the errors. (b) The slide with correct answers when students finish group discussion. Errors are bolded in red, and correct answers are bolded in green to give students easier access to the mistakes.
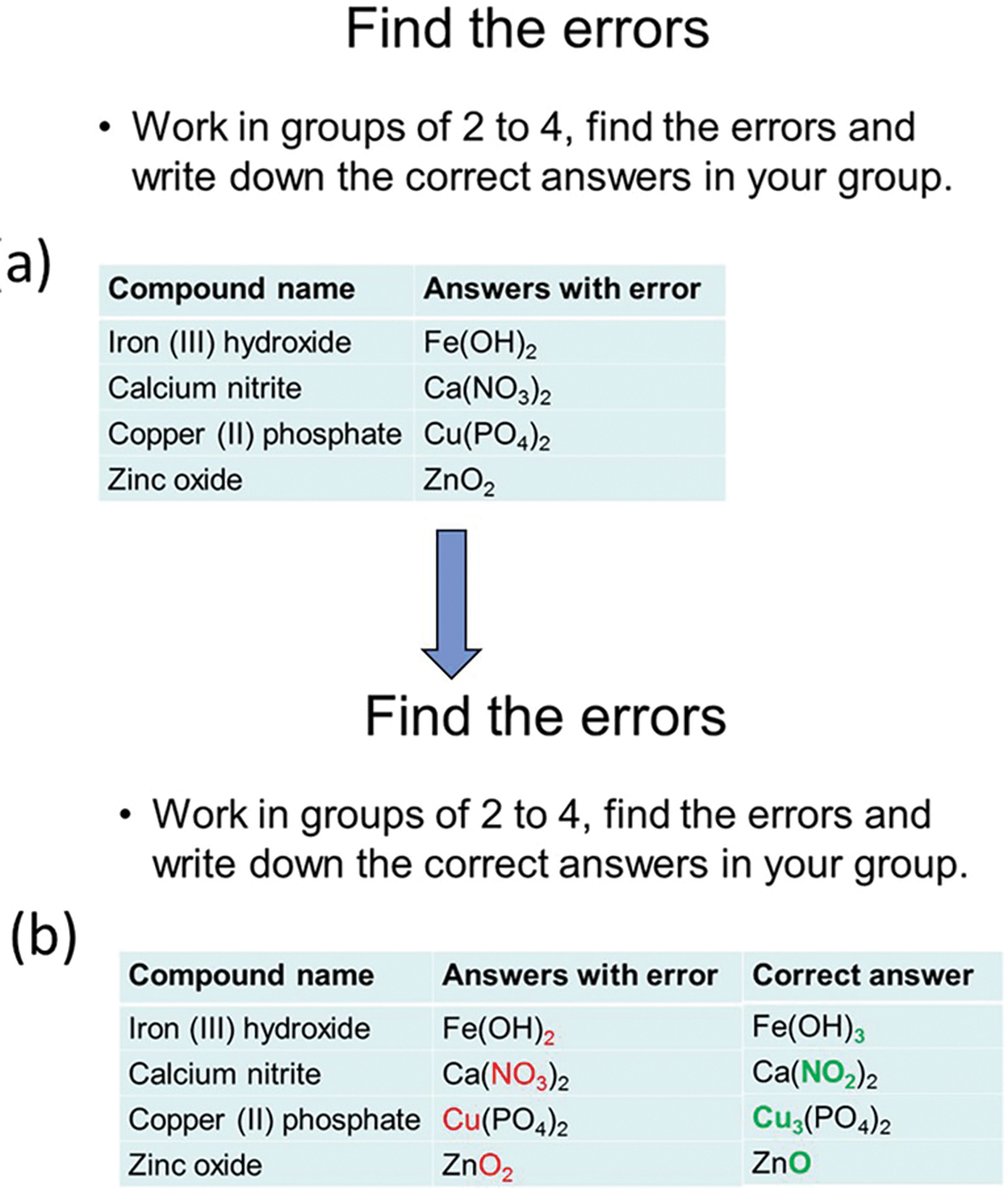
(a) The slide after the topic “Writing Ionic Compound Formulas” is taught. Students will work in groups to find and correct the errors. (b) The slide with correct answers when students finish group discussion. Errors are bolded in red, and correct answers are bolded in green to give students easier access to the mistakes.

(a) The slide after the topic “Writing Ground State Electron Configuration” is taught. Students will work in groups to find and correct the errors. (b) The slide with correct answers when students finish group discussion. Errors are bolded in red, and correct answers are bolded in green to give students easier access to the mistakes.
Observations/results
With the constructive error climate, students are encouraged to deal explicitly with the errors. Like many other CATs (Cross & Angelo, 1993), using errors as integral elements of the learning process is simple and effective, and some positive outcomes have been observed. During the class time, all students were engaged, focused, and following the instructions. The instructions were: Recognize the errors in small groups, figure out the reasons behind the errors, compose the correct answers and share between the groups. Most students were very happy to find an error and replace it with a correct answer, and they requested more of such activities. After the first few weeks of the course, it was observed that students gained more confidence in their problem-solving ability and were more likely to share their thoughts during class, which helped develop their higher-order thinking skills. Also, in the small groups (Wilson & Varma-Nelson, 2016), students were stimulated and promoted to speak up in front of their classmates.
This CAT also enhances awareness of how students understand the material, and the instructor learns notable information about students’ learning while students work on the errors. When misconceptions in understanding show up (e.g., the same one error is not recognized by most students), then on-the-spot lecturing is introduced by the instructor so that those misconceptions can be addressed immediately, rather than later when they appear on homework and exams. It was observed that students’ average homework grades were improved by about 10% during implementation of this CAT.
Students have also shown appreciation for utilization of errors and their reactions toward this CAT were extremely positive. For example, numerous students have commented that they never realized that the errors could help improve their learning; they enjoyed learning with identifying errors, assessing errors, and giving insightful thoughts on improving their learning, and they had more understandings on topics when this CAT is implemented.
Summary
According to the theory of negative knowledge, a classroom with a constructive error climate is beneficial for students to learn from errors (Gartmeier et al., 2008). The CAT previously mentioned could contribute to a constructive error climate by using errors as opportunities for learning. The scope of a concept is sharpened by clarifying what does not belong to it, and students feel more confident dealing with errors.
Establishing a contrast between positive and negative knowledge requires students’ prior knowledge (Heemsoth & Heinze, 2014). Therefore, an activity incorporating errors should not be used to introduce a new concept. Moreover, after thoroughly reflecting on the errors between students, the instructor should provide students with the correct answers, such as the ones shown in Figures 1b, 2b, and 3b.
The benefit of learning from errors has not been realized in teaching, as correct examples are popularly used in classrooms. Therefore, more research-based study implementing this approach is still needed.
Yunteng He (yuntenghe@cccneb.edu) is a chemistry instructor at Central Community College in Kearney, Nebraska.
Assessment Preservice Science Education Research Postsecondary


