Research & Teaching
Cross-Disciplinary Learning
A Framework for Assessing Application of Concepts Across Science Disciplines
Journal of College Science Teaching—September/October 2022 (Volume 52, Issue 1)
By Emily Borda, Todd Haskell, and Andrew Boudreaux
We propose cross-disciplinary learning as a construct that can guide instruction and assessment in programs that feature sequential learning across multiple science disciplines. Cross-disciplinary learning combines insights from interdisciplinary learning, transfer, and resources frameworks and highlights the processes of resource activation, transformation, and integration to support sense-making in a novel disciplinary context by drawing on knowledge from other prerequisite disciplines. In this article, we describe two measurement approaches based on this construct: (a) a paired multiple choice instrument set to measure the extent of cross-disciplinary learning; and (b) a think-aloud interview approach to provide insights into which resources are activated, and how they are used, when making sense of an unfamiliar phenomenon. We offer implications for program and course assessment.
Amara is beginning her second term at university. She hopes to become an engineer and has thrown herself enthusiastically into her coursework. Despite this effort, she struggled in first-semester physics and chemistry. In her second chemistry course, she is learning about exothermic and endothermic reactions. When the class discusses the combustion of methane as an exothermic reaction, Amara is puzzled that energy seems to come from nowhere. She remembers the law of conservation of energy from physics and thinks, “The energy must come from somewhere, but where?” Her professor says something about energy being stored in the methane, which makes Amara think of potential energy, another idea she studied in physics. She recalls an example of a skateboarder riding down a hill, during which potential energy was transformed into kinetic energy. She wonders if potential energy is transforming as methane burns, but she is not sure what potential energy is for methane or what other type of energy it becomes during its transformation.
Miguel is in the same second-term chemistry course. An aspiring biochemist, he studied hard during his first term and earned top grades in both physics and chemistry. Now, during the class discussion of methane combustion, he writes down notes and definitions, highlights words such as “enthalpy,” and memorizes heuristics such as “exothermic = exit” and “endothermic = enter.” He launches into learning these new facts. “Energy again,” he thinks. “Enthalpy must be a new type of energy for chemistry. Glad we’re not talking about that potential energy stuff anymore.”
Although these characters are fictional, they are likely familiar to many instructors. While Amara exhibits an approach to learning we hope to instill in our students, Miguel’s approach may be more likely to lead to academic success. We aspire for students to seek connections between disciplines, but we recognize that common academic structures and assessment practices, including those at our institution, may reward students who focus narrowly on the content associated with a single course.
Here we propose the construct of cross-disciplinary learning, drawing on several well-established theoretical frameworks, and describe two associated assessment approaches. We present these approaches as tools for making cross-disciplinary learning visible so that it can be promoted at the classroom, departmental, and interdepartmental levels. Elsewhere, we detail the development and validation of the instruments and protocols and present a comprehensive analysis of data they have generated (Haskell et al., 2022). In this article, we limit ourselves to describing our approach, in the hope of stimulating discussion about how to see, reward, and further develop habits of mind like those of Amara.
Significance
The sequenced structure of many undergraduate science programs is consistent with two assumptions about learning: first, that concepts and skills acquired in earlier courses serve as building blocks for mastering content in later courses, and second, that students will productively combine those concepts and skills to support their learning in new disciplinary contexts. Shared concepts can be used to bridge knowledge from different science disciplines. For example, the general rule that the potential energy of a system of attracting objects increases with object separation can be used to make sense of why a ball slows down when thrown upward and why energy input is needed to break a chemical bond. While students could reason by analogy directly from one situation to the other (Gentner, 1989), knowledge of shared concepts will help them generalize to other situations involving attracting objects, regardless of the discipline.
Our work has focused on energy, a fundamental framework for building explanations in all science disciplines (Eisenkraft et al., 2014). Energy’s importance is highlighted as both a core idea and a crosscutting concept in the Next Generation Science Standards (NGSS Lead States, 2013). The siloed nature, distinct foci, and technical vocabulary of the disciplines, however, can obscure the universal applicability of energy (Eisenkraft et al., 2014). Furthermore, as illustrated in the opening vignette, assessments may not be sensitive to student learning of concepts that cut across disciplines.
The insights driving our approach originated from our work in a coordinated science content course sequence for preservice elementary and middle school teachers, including one course each in physics, geology, biology, and chemistry. The curricula (Donovan et al., 2008; Goldberg et al., 2005; Goldberg et al., 2016; Borda et al., 2014; Borda et al., 2017) all focus on the flow of matter and energy; use similar vocabulary, representations, and modeling tools surrounding energy concepts; and employ a common, constructivist-based pedagogy (Baviskar et al., 2009; Colburn, 2000). The coherent design of this series has made it a valuable laboratory for exploring students’ cross-disciplinary learning of energy. Common themes and pedagogies provide opportunities for students to make connections across disciplines, and they make crosscutting concepts more visible than they might be in traditional science courses (Acevedo-Gutierrez et al., 2016; Donovan et al., 2013). For example, students develop and use energy diagrams—a specific representation—throughout the series and use common vocabulary around energy forms and transfer types. We hoped this course series would allow us to see, characterize, and measure cross-disciplinary learning.
Although our work in this instructional setting has played an important role in the development of the assessment approaches described in this article, we recognize that a siloed structure remains the norm in science, technology, engineering, and mathematics (STEM) education, including at our institution. We believe that the assessment tools we have developed for characterizing cross-disciplinary learning are appropriate for use in traditional, more siloed course sequences in which concepts are presented in the context of a particular discipline, without explicit discussion of how those concepts might also be used in other disciplines. It is perhaps in that context where making cross-disciplinary learning visible is most valuable.
Conceptualizing and measuring cross-disciplinary learning
In developing and teaching the course series mentioned above, we increasingly felt the need to gauge how well the courses were helping students apply energy ideas across different disciplinary contexts. Achieving this goal required a theoretically grounded construct that foregrounds the process of making connections. In this article, we draw on existing theoretical approaches to help define cross-disciplinary learning. We then apply this construct to describe novel approaches to assessment.
Defining cross-disciplinary learning
The vignette featuring Amara illustrates the kind of science learning we hope our students will achieve. In this vision, concepts and skills acquired in foundational courses are combined to support learning in later courses, including in novel disciplinary contexts. We refer to this process as “cross-disciplinary learning” and provide a model, featuring Amara’s reasoning, in Figure 1. Encountering a learning challenge in a novel disciplinary context triggers activation of knowledge developed in previous coursework, such as ideas about potential energy and energy conservation from Amara’s physics course, as well as knowledge from current coursework, such as the observation of increasing temperature during methane combustion. Some activated knowledge elements are transformed as Amara combines them. For example, the idea that potential energy transforms into kinetic energy, originally learned in physics, is recast in a more general form, which can be more readily combined with the knowledge from chemistry that heat is released when methane combusts. The integration of these energy ideas then supports the development of a new insight that potential energy associated with methane transforms during combustion. Overall, cross-disciplinary learning involves active synthesis, possible through the (perhaps implicit) recognition that some ideas (e.g., energy cannot be created or destroyed) apply to situations with widely disparate surface features (e.g., skateboarding and methane combustion). We note that activation, transformation, and integration are broadly consistent with constructivism (Colburn, 2000; Piaget, 1978; Vygotsky, 1978), which frames learning as an active, social process in which learners develop ideas based on experience and evidence, rather than a passive assimilation of knowledge.
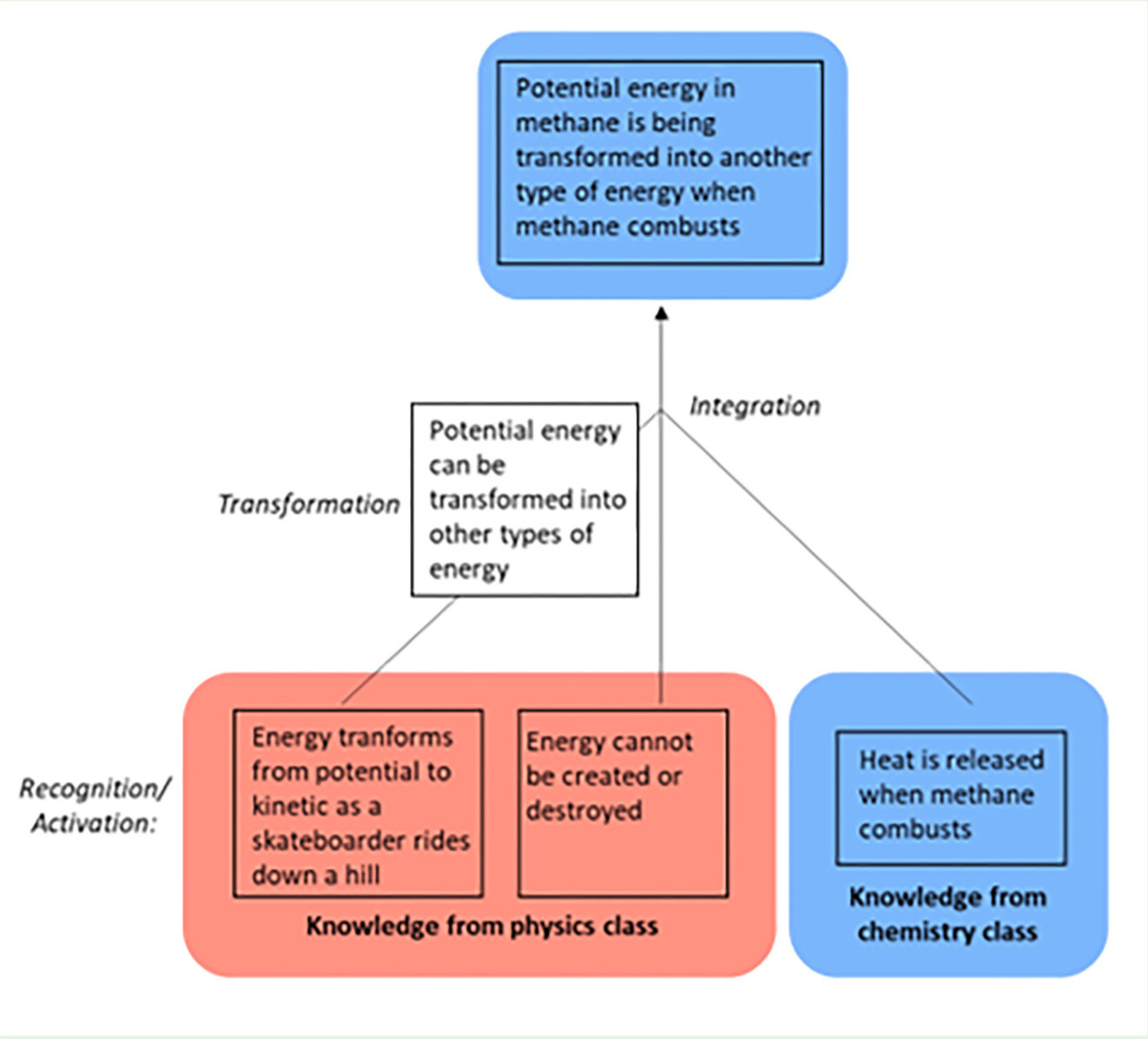
A model for cross-disciplinary learning.
To support and further elucidate this model for cross-disciplinary learning, we make connections with existing relevant theoretical frameworks, including interdisciplinary learning, transfer, and resources. We describe how each of these relates to cross-disciplinary learning and illustrate how this novel construct can provide guidance for developing assessments that will be sensitive to the type of sense-making exhibited by Amara.
Interdisciplinary learning
Shen et al. (2015) propose an Interdisciplinary Reasoning and Communication (IRC) framework for how individuals leverage knowledge from multiple disciplinary contexts to solve a complex problem. Two features of this framework are transformation and integration, which play a central role in our model of cross-disciplinary learning. Transformation involves changing the form of a knowledge element to enhance its applicability to a new context, while integration involves combining different elements.
Examples of transformation we have observed in student reasoning include generalizing from gravitational interactions to other types of attractive interactions (e.g., magnetic, electrostatic) when reasoning about potential energy changes and extending the concept of kinetic energy from the context of a single macroscopic object to the context of submicroscopic particles when reasoning about temperature changes in a chemical system. Examples of integration include connecting the concept of potential energy to the idea of adenosine triphosphate (ATP) as an energy carrier and combining a small particle model of matter with the notion of heat transfer to develop particle collisions as a mechanism for temperature changes.
Interdisciplinary learning frameworks tend to foreground a problem or context and seek to identify the different ideas students combine in finding solutions (Shen et al., 2014). Such frameworks are especially applicable for integrated science or problem-based learning curricula. Our focus on sequential learning is somewhat different from the problem-based approaches described in some interdisciplinary learning frameworks. Although interdisciplinary learning and cross-disciplinary learning do overlap, our formulation of cross-disciplinary learning shifts focus toward the new ideas and knowledge students develop and away from the problem or context designed to spur sense-making.
Transfer
Transfer frameworks are relevant to cross-disciplinary learning in part through their shared focus on sequential learning. Two specific kinds of transfer are relevant to our conception of cross-disciplinary learning: direct transfer and Preparation for Future Learning (PFL). Direct, or classic, transfer research has examined the extent to which students apply learned concepts, reasoning, or problem-solving skills directly to a new target domain (e.g., Gick & Holyoak, 1983). Traditional assessment methods often fail to detect direct transfer when there are many differences between the learning and application contexts—so-called “far transfer” (Barnett & Ceci, 2002). Dreyfus et al. (2014), for example, describe a student struggling to adapt the idea of potential energy stored in gravitational fields to potential energy associated with chemical bonds to explain energy changes during chemical reactions. The gravitational field conception does not support the use of potential energy in an explanation for chemical reactions and thus cannot be transferred directly to leverage this explanation. As this example illustrates, cross-disciplinary learning often requires transformation of concepts for productive connections to be made. Thus, while we see direct transfer as an important theoretical idea to draw on, for our purposes it was important to foreground the integrative and transformative processes involved in cross-disciplinary learning.
In contrast to direct transfer, in which a learned concept or skill is applied “as is” to a novel challenge, PFL frames prior learning experiences as preparation for an active process of constructing knowledge in the new context (Bransford & Schwartz, 1999; Schwartz et al., 2005). This view has clear overlap with cross-disciplinary learning: Knowledge developed in a prerequisite course provides students with tools for reasoning and learning in a new context. For example, familiarity with energy conservation from physics led Amara to ask questions about what forms of energy are changing and how potential energy may be involved when learning about combustion in chemistry.
Resources
To transform and integrate knowledge from different disciplines, Amara must first activate relevant knowledge learned in those contexts. Conceptual resource frameworks describe small-grained, existing bits of knowledge that are marshalled to form larger ideas or to explain a phenomenon (Benedikt et al., 2013; Richards et al., 2018). Resources related to energy include associating an energy form with a physical indicator and accounting for energy quantities before and after a process (Sabo et al., 2016). Resources function as building blocks of knowledge that are activated in context-specific ways. Because learning is defined in terms of the activation of existing cognitive entities, learning and transfer are not strongly distinguished in a resources framework (Hammer et al., 2005).
Combining these frameworks, we define cross-disciplinary learning as a process of sequential learning in which students activate, transform, and integrate knowledge from different disciplines, combining previous learning with new learning to construct new knowledge within a specific discipline. We propose cross-disciplinary learning not as an alternative to these theoretical frameworks, but rather as a combination of their elements that captures the learning in which traditional science programs seem to expect students to engage. The construct of cross-disciplinary learning is a useful guide when evaluating where that kind of learning is occurring and where it is not.
Measuring cross-disciplinary learning
Traditional, siloed science instruction relegates most or all responsibility for making connections between disciplines to the student. By offering approaches to measuring cross-disciplinary learning, we hope to support instructors in actively promoting it for their students, thus shifting to a model of shared responsibility. The approaches to assessment we describe could be used to generate formative feedback, at a classroom level, that could help faculty adjust instruction in real time. Assessments could also be used for more summative, program-level evaluation, which could spur collaborative work toward more coherent, integrated curricula. We offer both quantitative and qualitative approaches. While quantitative assessment provides insight into the extent to which different conceptual resources are activated, a qualitative approach can reveal what resources are marshalled and how they are transformed and combined.
Quantitative measurement of cross-disciplinary learning
Quantitative instruments can be used at the classroom level at the beginning of a term to help guide instruction, at the end of the term to show growth, or at the program level to motivate curricular changes. Several instruments exist to measure students’ understanding of energy in multiple science disciplines (Herrmann-Abell & DeBoer, 2014; Opitz et al., 2017; Park & Liu, 2016). However, to assess the more sequential construct of cross-disciplinary learning, we sought an approach that would be sensitive to students’ ability to use energy ideas to “figure things out” in an unfamiliar context, while backgrounding their ability to apply those concepts within each discipline. We also wanted assessments to be practical to use to inform teaching or program improvement. These considerations led us to develop a pair of instruments that assess a single energy idea—the conservation principle—in two disciplinary contexts: one that would be familiar to students and one that is more novel. We chose physics as the familiar context because it is a prerequisite in the course series that has guided this work, and we chose chemistry as the unfamiliar context because most of our students take chemistry last in the sequence. In many universities, including our own, students take physics as a prerequisite for science programs. Finally, students’ difficulty translating energy ideas from physics to chemistry is well documented (Becker & Cooper, 2014; Dreyfus et al., 2014; Lindsey et al., 2019; Nagel & Lindsey, 2015), establishing a need for tools to support cross-disciplinary teaching and learning in this area.
To illustrate the design of the instrument pair, we show a sample question from each instrument in Figure 2. Both questions involve the energy conservation principle, but the first question is in a physics context, a child throwing a ball, and the second is in a chemistry context, an electron moving in a Bohr atom. (Note that we are less concerned about the sophistication or currency of this model of the atom than we are about the students’ ability to reason with it.) Items from the instrument featuring the familiar disciplinary context “screen” for the activation of a conceptual resource. The physics question, for example, requires the idea that the kinetic and gravitational potential energy associated with projectile motion will obey “accounting rules.” Items in the less familiar context require a student to transform this conceptual resource and integrate it with knowledge specific to the new context. For example, in the chemistry question, the accounting resource must be integrated with knowledge that in the Bohr atom, an electron can “jump” to a higher shell, which corresponds with greater separation from the attracting proton and, therefore, higher potential energy.
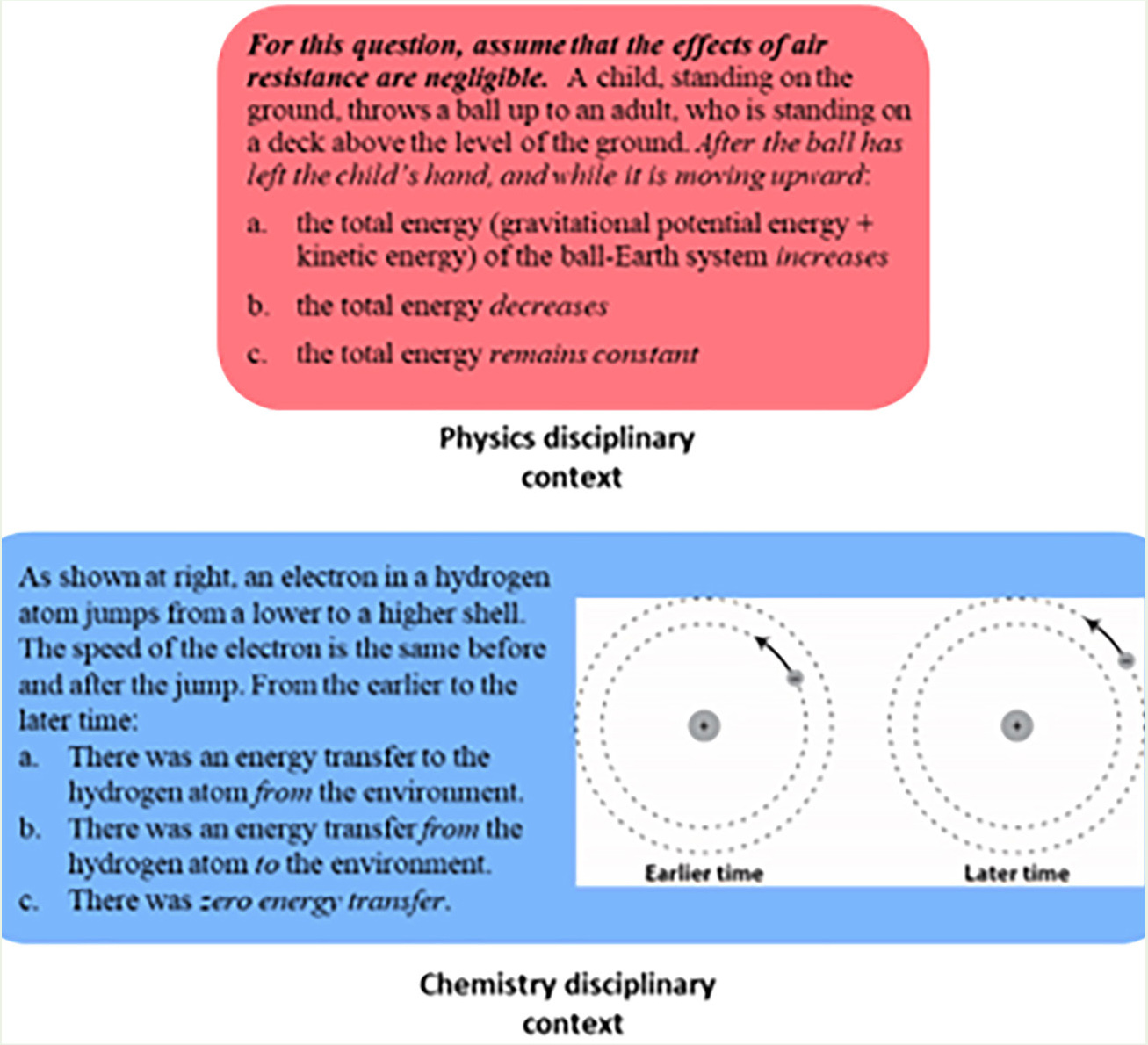
An example of an item pair for assessing cross-disciplinary learning.
For performance on this instrument pair to depend on student ability to reason across disciplinary boundaries, we wanted students to have sufficient information about the novel context to reason with. The chemistry items are grouped into four contexts: an electron moving in a Bohr atom, two ions forming a bond, phase changes in a pure substance, and chemical reactions. Each context is preceded with narrative text that introduces relevant energy forms and indicators. These narratives are followed by unscored reading comprehension questions, which give students feedback to help them refine their ability to use these new resources. The narratives and reading comprehension questions are built into the assessment instrument itself in an effort to foreground student reasoning (e.g., transformation and integration of concepts), rather than technical understanding of new vocabulary. Figure 3 shows the narrative and reading comprehension questions that precede the Bohr model items.

An example of a teaching narrative followed by two reading comprehension questions.
Some instructors may have concerns about providing chemistry content as part of the assessment. However, this measurement approach is not intended to measure student understanding in either of the disciplines, but rather students’ skill in leveraging their knowledge from one discipline to figure out new knowledge in the other (we administered the two instruments after physics instruction but before any relevant chemistry instruction). These instruments do not replace concept inventories, which can measure students’ understanding of relevant concepts as instruction proceeds.
The paired instruments share a common requirement for success: the activation of conceptual resources for energy conservation. The chemistry instrument adds the requirement that students integrate the energy conservation resources learned in physics with the newly introduced conceptual elements from chemistry. To describe a student’s ability to reason across disciplinary contexts, an instructor can compare their relative performance on the physics and chemistry instruments. Strong relative performance on the chemistry instrument suggests the student is able to combine already learned knowledge, as measured by the physics instrument, with the new information provided about the unfamiliar context. Relatively weaker performance on the chemistry measure suggests more compartmentalized learning.
Importantly, the extent to which such an instrument pair measures cross-disciplinary learning depends on the extent to which the second context is unfamiliar. For example, when faced with the chemistry items, some students may draw on knowledge gained in a high school chemistry class, rather than (or in addition to) the more recent learning from college physics. The extent to which the second context is unfamiliar for the target population must be addressed during validation of the instrument pair. Notably, our preliminary results show students having taken college-level physics but not college-level chemistry do better on the chemistry instrument than do students who have not taken any college-level science courses and that upper-division physics students do better on the chemistry instrument than upper-division chemistry students. In Haskell et al. (2022), we present the instrument pair in detail, describe its validation, and share results. Arguably, results on a paired assessment of this type have the potential to inform course- or program-level improvement regardless of whether or not students bring earlier learning experiences to bear in responding to the questions.
During administration, students complete the chemistry items before completing the physics items. This ensures the physics instrument does not itself act as a treatment, by cueing students to apply particular energy ideas, but rather measures the extent to which students understand the energy ideas they were trying to apply. The paired instrument approach can be used for program-level assessment, while instructors can use a paired question approach, perhaps asking for written follow-up explanations—on clicker questions, exit slips, quizzes, and exams—to formatively and summatively assess students’ cross-disciplinary learning in their courses. A paired question approach can also support discussions about, and seed expectations for, cross-disciplinary learning.
Qualitative measurement of cross-disciplinary learning
Qualitative assessment can provide rich depictions of how conceptual resources are combined and transformed as a student formulates an explanation in a novel disciplinary context. A qualitative approach is particularly well suited for formative assessment because knowledge of how students use conceptual resources can help the instructor guide students to connect new material to previous learning. In parallel with the quantitative approach, we developed a set of interview protocols for students in the science education sequence. The protocols were designed for use with students who have completed the physics course and just started either the geology, biology, or chemistry course. These students had developed energy concepts in a physics context but had not yet started to systematically apply those concepts in the subsequent discipline.
To elicit student thinking, we used a Think-Aloud Interview (TAI) approach (Bowen, 1994) and asked students to construct a scientific explanation for an observed phenomenon. Each protocol involved three scenarios: two for the less familiar disciplinary context (geology, biology, or chemistry) and one for the more familiar physics context. The same energy concepts are relevant for all three scenarios. As in our quantitative assessment, we present the physics scenario at the end of the interview so the interview itself does not cue students to apply relevant concepts in the novel discipline. For consistency, we have shown our chemistry protocol as an example in Figure 4.

Sample interview protocol used for qualitative assessment of cross-disciplinary learning.
The interview protocols are semi-structured, and each interview required 30 to 50 minutes to complete. Students were presented with a sequence of questions from the protocol. The interviewer deployed follow-up questions as needed to further draw out student thinking. Interviews were recorded using a Smart Pen. Participants were recruited from science education courses and compensated with gift cards. All provided informed consent.
Data collection and analysis thus far suggest this approach is useful for exploring what resources students activate and in what combinations, as well as how the resources are used in making sense of novel scenarios. A detailed description of this work will be the subject of a future manuscript. The basic structure of the interviews, involving open-ended questions that pair two contexts using parallel ideas, could be modified as exit slips or homework for course-level formative assessment. The interviews could also be conducted with a sample of majors or alumni as part of a summative program assessment plan.
Conclusion
We have proposed the construct of cross-disciplinary learning and have begun to operationalize it by outlining frameworks for measurement. We have focused our discussion on energy in physics and chemistry, but the approach could be adapted to other crosscutting concepts and disciplinary contexts as well. Paired multiple-choice items and cross-disciplinary think-aloud interview tasks can foreground resource activation and integration while reducing the probability that context-specific procedural or declarative knowledge (or its absence) will solely determine the quality of student responses.
We do not suggest discipline-specific learning outcomes and assessments be replaced or abandoned; rather, we argue assessments that focus narrowly on a single disciplinary context can be supplemented with approaches that explicitly target ways of making connections across disciplines. Such assessments could reward and encourage the type of thinking exhibited by Amara and motivate and assess the creation of curricula and pedagogies that would support students like Miguel in moving toward more integrative habits of thinking.
Developing assessments and pedagogy focused on cross-disciplinary learning requires collaboration between disciplinary units to determine learning outcomes, frame them with common vocabulary, and develop curricular coherence. This may be especially true in introductory coursework, in which crosscutting ideas come up frequently but may be obscured from students by discipline-specific vocabulary and representations. Perhaps most of all, prioritizing cross-disciplinary learning challenges us to think of ourselves as science educators first and disciplinary experts second. We hope this work supports conversations about how to envision coherent teaching and learning across our science programs.
Acknowledgments
This work was supported by the National Science Foundation (DUE-1612055, DUE-1612251). Any opinions, findings, conclusions, or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation. We are grateful to co-PI Sarah Julin for helping develop interview protocols and methods and to advisory board members Drs. Jack Barbera, Amy Robertson, and Ben Geller for feedback, advice, and guidance in conceptualizing cross-disciplinary learning and its measurement. We would also like to thank Drs. Deborah Donovan and Susan DeBari, who gave feedback on initial drafts of instruments and protocols, and Dr. Daniel Hanley, who provided formative evaluation in early stages of this project.
Emily Borda (bordae@wwu.edu) is a professor in the Departments of Chemistry and Science, Math, and Technology Education (SMATE), Todd Haskell (haskelt@wwu.edu) is a former professor of psychology, and Andrew Boudreaux (boudrea@wwu.edu) is a professor of physics, all at Western Washington University in Bellingham, Washington.
Crosscutting Concepts Curriculum Interdisciplinary


