Middle School High School | Daily Do
Why Is Water Sphere-Shaped in Space?

Aerospace Careers Chemistry Crosscutting Concepts Disciplinary Core Ideas Is Lesson Plan NGSS Phenomena Physical Science Science and Engineering Practices Three-Dimensional Learning Middle School High School Grades 6-8 Grades 9-12
Welcome to NSTA's Daily Do
Teachers and families across the country are facing a new reality of providing opportunities for students to do science through distance and home learning. The Daily Do is one of the ways NSTA is supporting teachers and families with this endeavor. Each weekday, NSTA will share a sensemaking task teachers and families can use to engage their students in authentic, relevant science learning. We encourage families to make time for family science learning (science is a social process!) and are dedicated to helping students and their families find balance between learning science and the day-to-day responsibilities they have to stay healthy and safe.
Interested in learning about other ways NSTA is supporting teachers and families? Visit the NSTA homepage.
What Is Sensemaking?
Sensemaking is actively trying to figure out how the world works (science) or how to design solutions to problems (engineering). Students do science and engineering through the science and engineering practices. Engaging in these practices necessitates that students be part of a learning community to be able to share ideas, evaluate competing ideas, give and receive critique, and reach consensus. Whether this community of learners is made up of classmates or family members, students and adults build and refine science and engineering knowledge together.
Introduction
Did you know humans have been living in space for the last 20 years? NASA is currently celebrating the Year of Recognition, highlighting the anniversary of 20 years of continuous human presence aboard the International Space Station on November 2, 2020.

Fluids—gases and liquids—are everywhere in our lives. The Earth, known as “the water planet,” is able to support life in part because of the presence of water. Understanding the basic principles of fluid motion, such as with water, is important for all walks of life. The behavior of water on the International Space Station (ISS) is not only mesmerizing and beautiful to observe (see picture below), but also researchers are using these observations to figure out why fluids behave differently in microgravity than they do on Earth. The near-weightless conditions aboard the ISS allow researchers to observe and control a wide variety of fluids in ways that are not possible on Earth. Understanding how fluids react in these conditions could lead to improved designs for space travel, as well as back on Earth.
In today's task, Why Is Water Sphere-Shaped in Space?, students and their families engage in science and engineering practices to make sense of the phenomenon of liquid water forming a perfect sphere-shape in space. Students see how the science ideas that explain water's sphere-shape in space are applied to the design of the Space Coffee Cup, then brainstorm solutions to other problems astronauts encounter that will help people live in space! While students could complete this task independently, we encourage students to work virtually with peers or in the home with family members.

Part 1: Why Does Water Form a Sphere in Space?
When people think of a liquid, they usually think of water. Students have many experiences with water and how it behaves on Earth. Water is so common and so familiar that it is mundane: Every day we drink it, touch it, wash with it, wet things, dry things, boil it, freeze it, and swim in it. We live in a world where the environmental conditions allow water to exist as a solid, liquid, and gas; where slight changes in temperature and pressure can cause water to alternate among phases (or sometimes be all three at once). The more we examine water, the stranger it gets. But few have had experiences with how it behaves in space!
Tell students, "We are going to watch this video of an interesting phenomenon in space!"
Ask them to make a simple T-chart on blank paper or in their science notebooks. Label one side "Notice" and the other side "Wonder," and ask them to record their observations and questions that arise as they watch the video.
Ask students to share their noticings with two or three other students (small group) and to identify (1) two or three observations everyone in the group had and (2) one observation that only one or two group members noticed. You may choose to create a class list of observations.
Note: Students have probably heard about water surface tension, but may not be able to use that science idea to explain why the water behaves the way it does on the ISS. If students don't bring up surface tension, that's okay.
Ask students to create an initial model to explain why the water squeezed from the pouch formed a sphere that the candy was able to stick to/sit on.
If students are struggling to begin, ask them, "What absolutely needs to be included in your model? In other words, what are the components of the model?" (water molecules, forces, molecular bonds, air, candy) Ask, "How could you show how the components of your model are interacting?" (arrows, lines, labels, text)
Working in pairs or small groups, ask students to compare their models and identify similarities and differences. You might ask student groups to create a consensus model. If you choose to do this, students can do a gallery walk. Ask them to use sticky notes to identify something on other groups' models that they like (maybe they like the way a group represented components interacting, for example) and one thing they have a question about. Allow the groups to add to or change their models after the gallery walk.
Before moving to the next investigation, give students an opportunity to think about related phenomena. These prompts may help elicit students' ideas:
- What experiences do you have that might help you think about this phenomenon?
- Is this something you’ve heard about, observed, or experienced before?
- What questions does this raise for you?"
Notes for Educators (Teachers and Families)
Here on Earth, we all live in a state of gravity. Not only us, but everything around us, including water, is being pulled toward the center of the planet by gravity.
But if you go far enough out in space, for instance, onto the International Space Station, gravity becomes negligible, and the laws of physics act differently than here on Earth. Just how might water act in a place of zero gravity? Actually, on the ISS, there is plenty of gravity. According to NASA scientists, the pull of Earth's gravity on the space station and its occupants is substantial: about 90% of the force at the Earth's surface. But since the space station is continuously falling around our planet, the astronauts and objects on board are in a kind of free-fall, too, and feel nearly weightless. Water on the space station behaves as if in a zero-gravity environment.
Part 2: Let's Explore How Water Behaves!
Tell students they are going to do a NASA investigation in their own home! Students and families will need to gather these household materials to see water behave in ways similar to how it does in space.

Materials
- A penny
- Small disposable cups (Plastic 7-oz. cups work great!)
- Water dropper (Rinse out any water dropper from old cosmetics or medicines, or make one from a straw.)
- Glass of water to fill cups and droppers
- Several grains of rice
- Pen/paper to record data
- Paper towels
Explore!
Let students explore without providing any explanation.
- Place the plastic cup on a paper towel and fill the cup to the very top with water.
- Add drops of water to the cup using the water dropper to see how high you can get the water above the rim without spilling over. (Record observations on paper using words, pictures, symbols, etc.)
- Now gently place a grain of rice on the water. (Record observations.)
- Next, take another grain of rice and throw it (give it a big push from your hand) onto the water surface. (Record observations.)
Set the cup aside and place a penny flat on a paper towel. Use the water dropper to carefully add water to the surface of the penny until it spills over. Try to keep the drops the same size. (Record observations.)
Discuss
Use the following prompts to spark discussion:
- How many drops did it take to overflow the cup or spill over the sides of the penny?
- Why do you think you were able to add water drops higher than the top rim of the cup or higher than the surface of the penny without it flowing over at first?
- Why do you think the first grain of rice stayed on the water surface?
- Why do you think the second grain of rice acted differently?
Tell students they can use the words surface tension to communicate their observations to other students and scientists. (At this point in the task, ask students to revise their models using evidence from these investigations.)
Notes for Educators
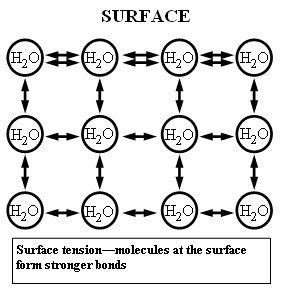
Surface tension is a property of liquids in which molecules of one substance are more attracted to one another than to molecules of another substance. Water is unique in that it has a high surface tension compared to other liquids. We observe this when water beads up on glass. Water molecules are able to form hydrogen bonds due to the polarity of the H2O molecule. The slightly positive ends of the hydrogen atoms and the slightly negative end of the oxygen atom attract other H2O molecules, forming a hydrogen bond, and causing water to have high surface tension. At water’s surface, its molecules are only attracted to the water molecules below and to the sides of them, as there is only air above these molecules. Therefore, the surface molecules of a body of water are pulled down, creating a much more stable, stronger arrangement.
More force is needed to break the water molecules' bond, which is why the second grain of rice broke through and sank to the bottom of the cup.
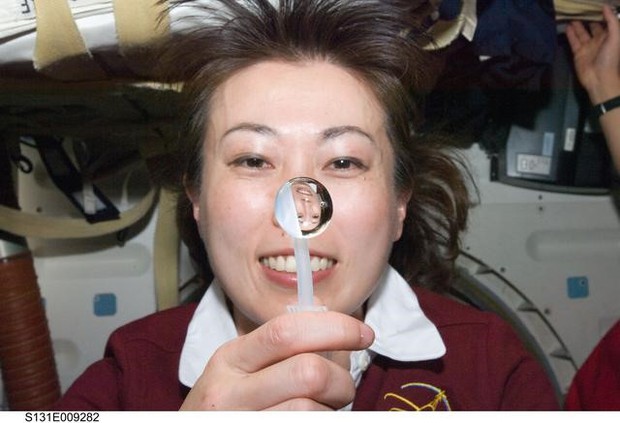
STEMonstration: Surface Tension!
Ask students the following questions to help navigate them to the STEMonstration: Surface Tension! video.
1. What would happen if you tried these experiments in microgravity?
2. Would the water behave differently in microgravity? Why or why not?
3. In microgravity, does surface tension exist? Why or why not?
Have students watch this episode of STEMonstrations, which features NASA astronaut Ricky Arnold explaining how water’s unique molecular bonding properties create quite the spectacle in microgravity!
Ask students to revise their individual or group models using evidence from the STEMonstrations: Surface Tension! video.
Ask students to find a partner (or groups to find another group) and share what they added to or changed on their model and why. (What evidence from their investigations or the video did they base their changes on?)
Lead the students in a consensus discussion. Consider using the following prompts:
- What are some things we think we can say about why water forms a sphere in space? What is our evidence for those ideas (those explanations)?
- What ideas are we in consensus about?
- Are there still places where we disagree?
You might choose to draw a consensus model, then have all students record the class consensus model on blank paper or in student notebooks. If you are working with middle school students, it is okay if they don't include molecular bonds in their models as long as they represent the fact that water molecules at the surface are more tightly together/have higher attraction to one another/etc.
Notes to Educators
How do we know surface tension in microgravity is no different than on Earth? The chemical bonding mechanisms for water molecules do not change in microgravity. These cohesive bonds are hydrogen bonds and are formed when the slightly positive (hydrogen) end of one water molecule is attracted to the slightly negative (oxygen) end of another water molecule, forming a series of water molecules that “stick” together and help produce water’s high surface tension. At the surface, water molecules are only attracted to other water molecules below and to the sides of them, and the inner cohesive forces are pulling these molecules together. Therefore, the surface molecules of a body of water are experiencing a net inward force, creating a much more stable, stronger arrangement. On the International Space Station, surface tension of water is much more obvious than on Earth due to the microgravity environment. What variables are acting on the water? One variable is the amount of gravity acting on the water.
Whether water sits in a lake or in a glass, Earth’s gravity pulls the liquid downward into the shape of the container it’s in. But in space, gravity’s effects are different. Objects in orbit are indeed affected by gravity, but they are in free-fall, moving constantly sideways while falling toward Earth. This renders them effectively weightless. Up there, surface tension among the tight-knit group of water molecules forms the smallest possible area: a sphere. On Earth, gravity distorts the shape, but not in space.

Part 3: Engineering a Good Life in Space!

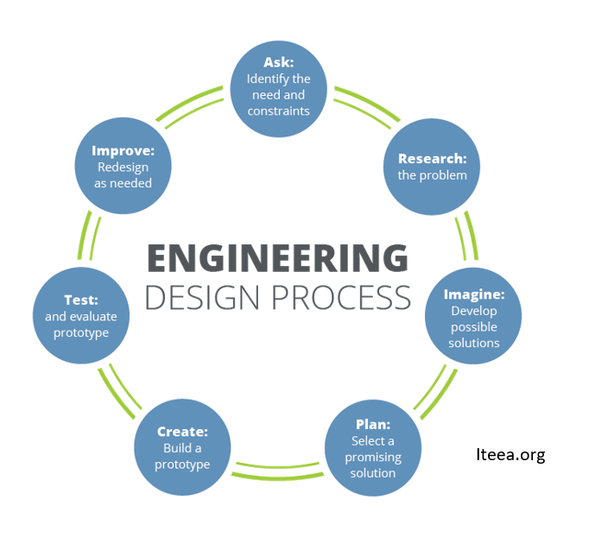
The Engineering Design Process (EDP) comes in many forms. Engineers enter the EDP to create a new technology—or improve an existing one—to meet a need or want. Engineers on the job may start at any step, depending on the needs of a particular project.
Share the The Zero Gravity Coffee Cup video with your students to find out how engineers helped astronauts drink a real cup of coffee in space. Ask students, "As you watch the video, can you identify the steps of the EDP used to design a solution to the problem of how can we drink a cup of liquid coffee in space? What's your evidence?"
Watch this video to learn about more challenges astronauts encounter in space. Does the video spark an idea for a new technology (object, system, or process) that would help make life in space easier and that you could design using the engineering design process?
Part 4: Explore STEM Careers: Astronaut Launch!
Share with students, "Now that you and the researchers figured out why water is sphere-shaped in space, explore what it takes to enter the exciting STEM career as an astronaut!"
STEM Career Awareness is an important part of educating and preparing our students for the future workforce. Students can explore the challenging and rewarding career of a NASA astronaut by watching and discussing these two videos.
How Did Leland Melvin Reach for the Stars?
Learn how Astronaut Leland Melvin, whom you met in the first video (playing with water and candy), went from NFL star to astronaut!
NSTA Collection of Resources for Today's Daily Do
NSTA has created a Why is Water Sphere-Shaped in Space? collection of resources to support teachers and families using this task. If you're an NSTA member, you can add this collection to your library by clicking Add to My Library, located near the top of the page (at right in the blue box).
Check Out Previous Daily Dos From NSTA
The NSTA Daily Do is an open educational resource (OER) and can be used by educators and families providing students distance and home science learning. Access the entire collection of NSTA Daily Dos.
Acknowledgments
This Daily Do is adapted from a NASA STEM demonstration or STEMonstration on Surface Tension. Please check the collection for the link and to explore other STEMonstrations to try!
Thank you, NASA Office of STEM Engagement at Johnson Space Center, for sharing your expertise and resources! stem.nasa.gov



