feature
Mud as a Phenomenon to Model Energy Flow and Chemical Processes
Science Scope—March/April 2021 (Volume 44, Issue 4)
By Hannah T. Reynolds, Joy Pires, Jeffrey Brewster, Nicole Lalier, and Theodora Pinou

CONTENT AREA Biology or Life Sciences
GRADE LEVEL 6–8
BIG IDEA/UNIT Changes in a Winogradsky column can be used to infer evidence of life and the cycling of matter and flow of energy into and out of an ecosystem.
ESSENTIAL PRE-EXISTING KNOWLEDGE The difference between living and nonliving things, how resource availability can affect growth
TIME REQUIRED Setup: 75 minutes; model construction and refinement: two 75-minute periods; multiple observation sessions: 10–15 minutes; final model construction and comparison: 75 minutes.
COST Gloves (100/box) $15
SAFETY Gloves and goggles for handling the mud and pond water; desks covered with paper or placemats before beginning
Sergei Winogradsky, a 19th-century microbiologist, developed a model ecosystem in a jar using water and pond sediment (Dworkin and Gutnick 2012). The Winogradsky column (see Figures 1 and 2) has been used to teach nutrient cycling and energy flow (Anderson and Hairston 1999; Rogan et al. 2005; Parks 2015). This article describes how middle school teachers used the Winogradsky column as a phenomenon to teach the biochemical processes of life (photosynthesis and respiration) and energy flow in ecosystems. This safe and inexpensive activity integrates science and engineering model-building practices and lets students explore the cause and effect of light and oxygen on growing organisms. This activity can be conducted in the classroom or at home, and thus has the added value of supporting science-at-home remote learning.
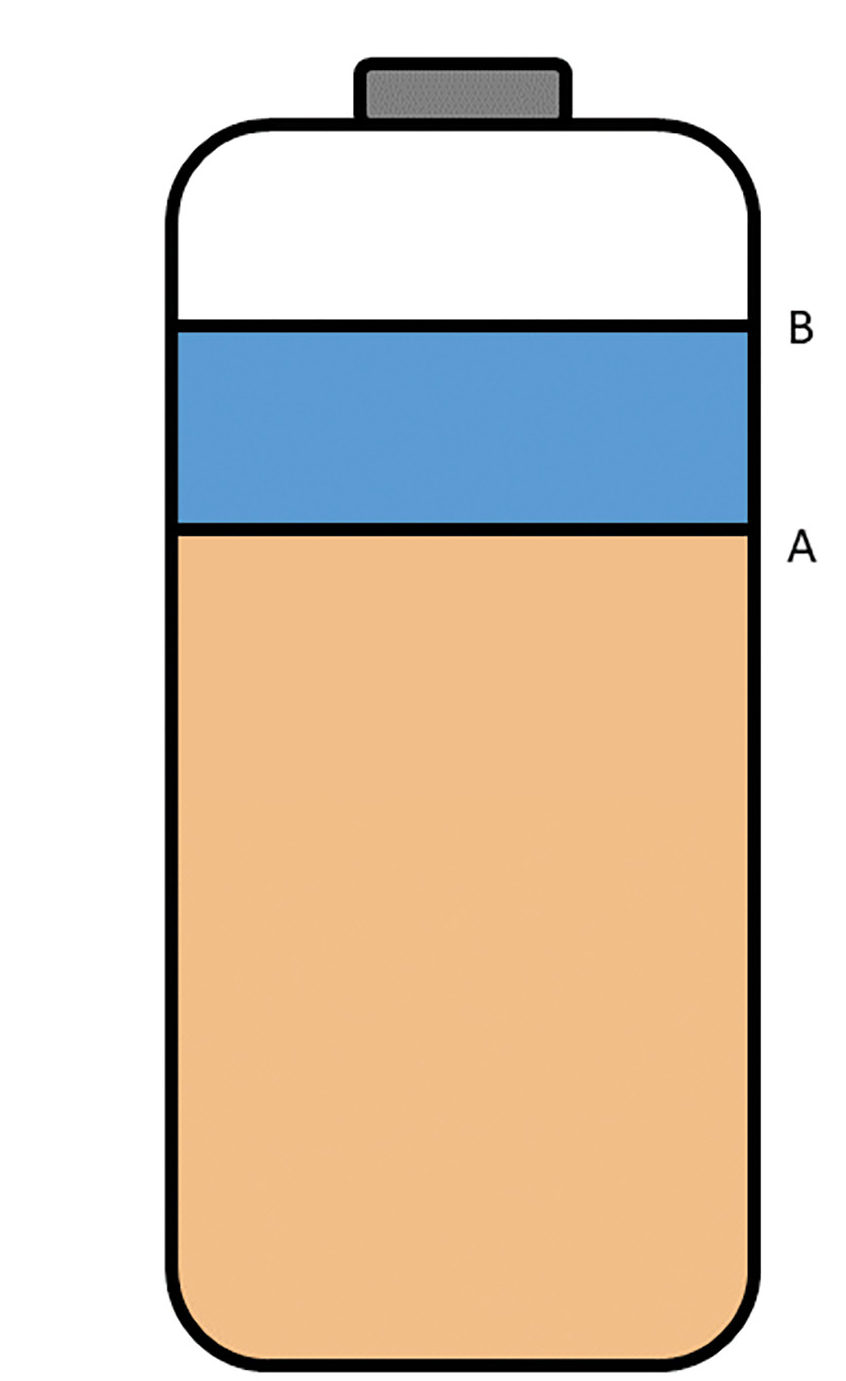
Winogradsky column diagram. Sediment fills 2/3 of the bottle (line A), and water is added to the 5/6 mark, leaving 1/6 of the bottle for air space. The bottle is loosely sealed or covered to permit gases to escape.
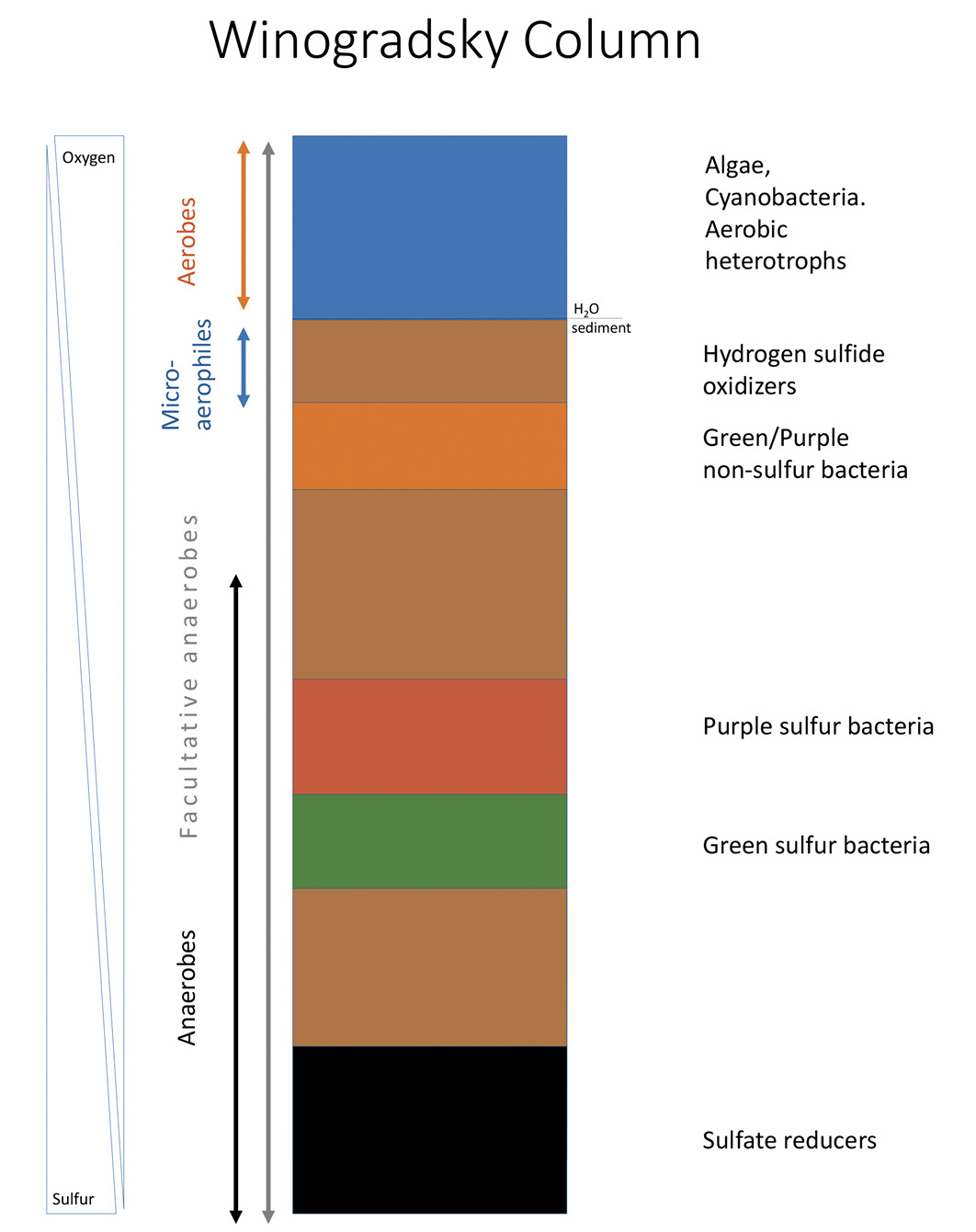
Winogradsky column microbial communities. Over time, microbial communities form colorful zones based on their oxygen and sulfur requirements. This diagram shows the organisms that will grow in the water and sediment portions of the column.
In a Winogradsky column, microbes in the mud and water replicate and form visible populations over the course of several weeks (Anderson and Hairston 1999). Light, soil chemistry, and oxygen are variables that affect the growth of organisms naturally occurring in pond water and/or mud, and this growth can be observed, identified, and measured. If aquatic seeds germinate and grow into visible plants, or animals such as worms and aquatic insect larvae move, life in the column becomes easy to observe. However, even in columns that lack obvious organisms, observable changes indicate life is present. Within a few days of setting up the Winogradsky column, bubbles will start creating a foam on the top of the water as a result of microbial metabolism. As bacteria multiply, they form splotches and bands in green, orange, purple-red, and black at distinct locations in the columns and are thus visible without a microscope (see Figure 2), similar to mold growing on a piece of bread. Only columns kept in the light will develop bands formed by photosynthetic bacteria containing chlorophyll (green) or bacteriochlorophyll (purple-red); these zones will form where light is most plentiful. In this class, we did not distinguish between aerobic and anaerobic respiration, but aerobic respiration occurs near the top of the column, whereas anaerobic respiration and fermentation drive microbial growth at the bottom of the column where oxygen is absent.
Implementing the activity
Seventh-grade teachers worked with a local university to develop activities using Winogradsky columns as a phenomenon for a six-week unit on cells. After a brief introduction to the cells unit, student groups set up Winogradsky columns and observed them every class session for two weeks (meeting three times a week for a total of six observations), followed by once a week for four weeks (four observations), for 10 observations total.
In preparation, the instructors collected approximately 20 clear 16-ounce plastic water bottles, removed the labels, and cut off the narrow tops using scissors to make it easier to add materials (the cutting step can be avoided if using wide-necked bottles). After estimating the volume needed for all class columns, the teachers collected pond water and sediment from a nearby pond in clean plastic bags and gallon jugs. These materials should be collected within a week of starting the experiment, with enough mud to fill 2/3 of the bottle, and enough water to fill half of the bottle. If pond materials are not available, possible replacements are filtered water with soil from a garden, lawn, or other natural area (but not potting soil or compost). To limit the mess, teachers put the mud needed into small sandwich bags for each group’s bottle. Using the newly collected mud and pond water, the instructors prepared an example column to help students visualize the final column.
In terms of instruction, the teacher should determine in advance where to store the Winogradsky columns and whether they want to have students make observations in individual or team notebooks. We recommend that notebooks be kept in the classroom so they are always available for use. Journaling for this activity can also occur in an electronic notebook.
Before class, student team stations were created with aluminum food trays (approximately 13” × 20” × 3”). They contained the materials that were needed for the activity: precut bottles, gloves, goggles, markers, rulers,
plastic wrap, and masking tape. Students used scientific nitrile gloves, but rubber gloves or plastic food prep gloves could also be used. Latex-free gloves should be made available in the event a student is allergic to latex. Pond water was dispensed into smaller pitchers for student teams (leftover water can be repoured into the teacher’s collection jug). Make sure a funnel is available, or work outside or over a sink, since it can get messy to dispense the water. During the activity, we kept the bags of mud and pitchers of water on a separate counter until students were ready for these materials.
Session 1 (Week 1): Column setup (full period, 75 minutes)
While wearing gloves and goggles, students worked in groups of three to four to measure and mark lines on their bottles to indicate the volume of the sediment and water necessary (see Figure 1). After checking the bottles for correct measurements, the instructor gave each student group a plastic bag of pond sediment and a pitcher with pond water. Students then added the sediment to the bottles with their gloved hands, gently shaking the bottle from side to side to settle the sediment as evenly as possible (see Figure 3). Students were advised to pick out any large twigs or rocks, which could trap air pockets and change the oxygen levels of the deeper layers. Next, they added the pond water, gently shaking the bottle to remove air pockets from the side of the column.

Winogradsky column setup. Students set up columns using trays to organize their work stations, then placed covered and labeled columns in the window.
They taped plastic wrap over the columns, which should be loosely covered. If the columns are capped, they should be loosely capped for safety, so that gases from microbial metabolism can escape without building up pressure in the bottles. If using lids, they should be loose to allow for exchange of gases in the column. Students wiped stray mud and water from their bottles and labeled them with team names using available masking tape and permanent markers. Finally, they removed their gloves and left them on the trays, then placed the columns on the classroom windowsill and cleaned the workspace (see Figure 3). Student groups should have assigned spots on the windowsill in case some columns receive more light than others, as this may be used to help build explanations from observations. The teacher control column should be kept with the class columns, but with an opaque paper bag or box preventing it from receiving light. The team journaled their observations for Day 1, including observations of the control. At this point, all columns looked the same and were cloudy, as fine particles take hours to settle. Students assisted cleaning their stations with water, paper towels, and sanitizer. For additional options for making Winogradsky columns, see Online Resources.
Session 2 (Week 1): Observations and making predictions (20 minutes)
In the next class session, teachers instructed the students to work individually on their science journals for 20 minutes, drawing their columns and writing predictions of what would happen over time. Students predicted nothing was alive in the mud. They based their predictions on observations that they couldn’t see anything moving and/or they didn’t think anything could survive in mud. Teachers guided the learning by asking students to record observations like colors, bubbles, or living organisms. Water in the columns can spill, so students were instructed not to pick up the columns, but rotate them carefully to view all sides. Students were also invited to gently waft the air above their covered columns to check for odors as mud often contains sulfur compounds.
Session 3 (Week 1): Group model (full period ~75 minutes)
Students individually spent about 10–15 minutes of class time observing their column and recording observations similar to the prompts from the first session (color, bubbles, organisms). Then, students met in their groups to practice “science talk” and consensus-building skills to discuss if their observations agreed with their predictions of whether life was present in the mud. Students shared their ideas and worked with their team to create illustrated posters to answer the questions: “Is anything alive in this mud? How can you tell?” These team posters can be posted on the wall of the classroom as a class reminder of the experiment and necessary observations.
Sessions 4–10 (Weeks 2–6): Brief Observations (10–15 minutes)
Students journaled for 10–15 minutes, recording observations in their notebooks. Although students could not see evidence of life in their columns on the first day, after one week they observed bubbles, changes in odor, and visible small worms or worm tracks. It is this observable change in the bottle that drives the phenomenological pedagogy inspiring students to answer the questions, “Where did all this life come from?” and “Why do I see it now but couldn’t see it before?” Student conversations regarding their observations of color changes, specifically the green color on the top layer, showed they thought it had something to do with photosynthesis. Students also noticed movement of worms and aquatic invertebrates, and they thought it had to do with something alive.
Session 7: (Week 3): Refining individual and group ideas (full period, ~75 minutes)
Students refined their ideas to better explain what was happening in this ecosystem. If the class meets less frequently, this refinement should occur after about two weeks of column development. Observant students noticed colored zone formation, and teachers used guided prompts to remind students to look for bubbles as evidence of gases such as carbon dioxide and oxygen and color changes as evidence of chemical reactions. Using this evidence and their knowledge of photosynthesis and respiration, students revisited the question of life in the column, aligned observations to their new knowledge, and revised their ideas to explain where this life came from, given that they never added anything to the container after the first day.
Regarding concept development, initially, student observations were vague; most students did not make connections that microscopic living things were in the mud, although they had learned about bacteria the year prior. Neither did they mention photosynthesis or cellular respiration. Student observation skills improved as they could identify evidence of life through bubbles, colored zones, growing plants and algae, and worm tracks. The students’ excitement also increased as they started to understand that cells were present in their columns. Questions addressed by students included: “If there are no visible plants or animals in some of the columns, what is making the gases that are forming bubbles? Why are parts of the column turning green, but only in the areas exposed to sunlight?” Three weeks into the experiment (around Session 8), students noticed that columns were bubbling, with some columns forming a foam on the surface. The teacher guided the learning to ask: “Could these gases be evidence of living organisms? Which gas could these bubbles be?” (Although it is impossible to identify the type of gas in each bubble without specialized equipment, we know that if respiration is occurring, it would produce carbon dioxide as waste, and if photosynthesis is occurring, it would produce oxygen.) Throughout their discussions, the instructors helped students access their previous knowledge and guide them in developing meaning to explain their observations.
Session 10 (end of Week 6, full period, ~75 minutes): Final assessment
After the final observations, students answered a prompt in which they described how a microorganism can grow in mud by describing the role that processes of photosynthesis and respiration play in energy flow through their columns (see Figure 4). Their final observations included detailed drawings that included the color banding in the mud, bubbles, growing algal strands, and organismal movement. Groups presented their final ideas to the class to discuss how their thinking changed over the course of the unit. Students concluded that the color green was evidence of photosynthetic organisms, that living organisms were present even in areas of the column where there was no light, and that cellular respiration was allowing for organismal growth.
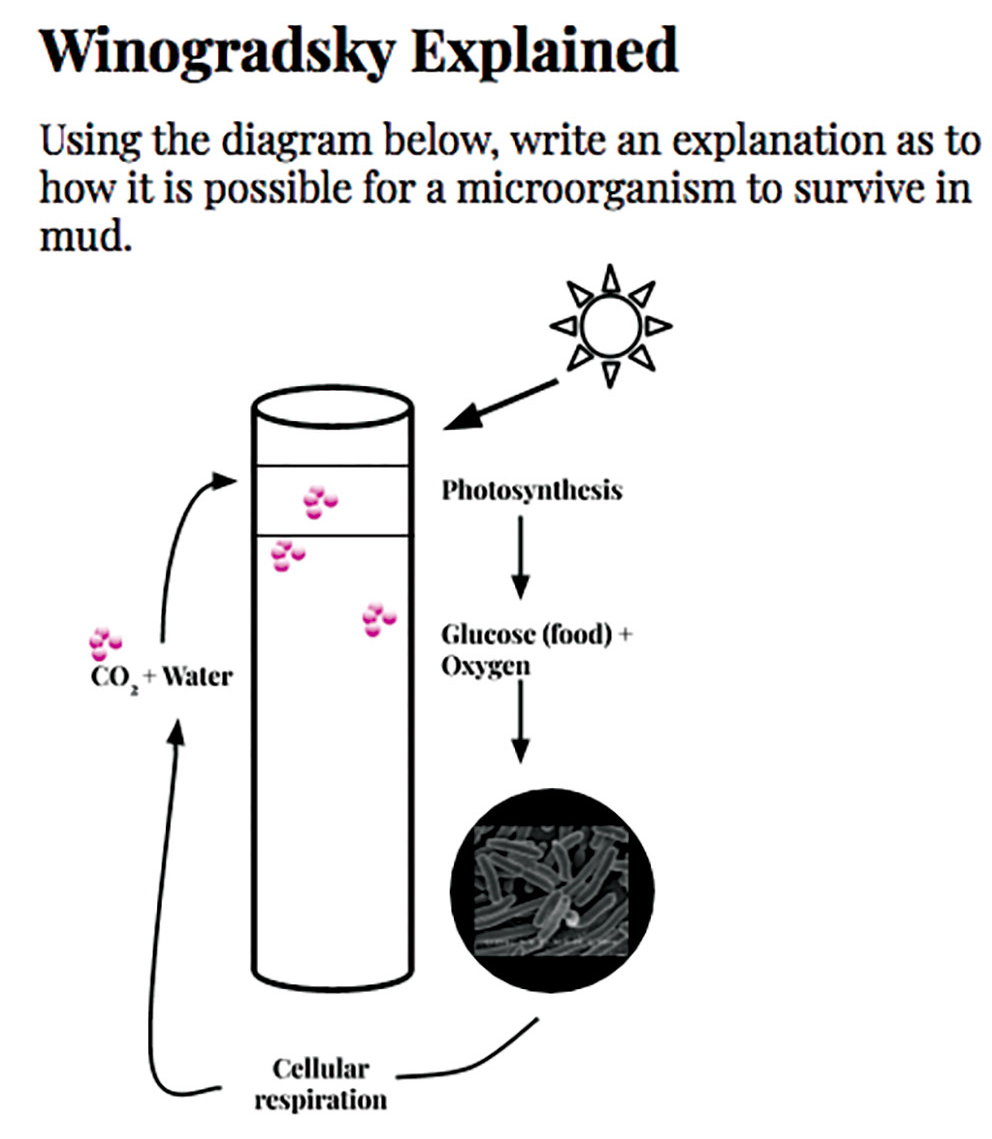
Final assessment. Students wrote a description of this diagram, explaining how a microorganism can grow in mud.
Regarding disposal, as long as no hazardous materials were added, the Winogradsky column contents can be returned to the original environment (if not from a protected habitat such as a nature preserve). Then the bottles can be cleaned and recycled. Otherwise, they should be thrown away as typical waste.
Adaptations and reflection
After performing this activity with students, we recommend that teachers make the activity more quantitative. Students could carefully measure the widths and depths of developing zones over time and compare mean values to the control. The students could also record the zone progress using either drawings or photographs. For a time series, it is critical to get a baseline, so students should record whatever they plan to observe (color zones, number of bubbles, odor, movement, etc.) on the first day, even if they have no data to record (i.e., “0” or “no smell”). At the end of the unit, students could compare each other’s observations using a gallery walk. As a final assessment, ask students to explain and provide evidence for how life can be present in a Winogradsky column.
Modifications for an online environment
The Winogradsky column experiment can be used as an at-home experiment. The instructor would need to guide students and parents on collecting the materials (bottle, ruler, markers, sediment, water). The parent may need to cut the top of the bottle for the student. The instructor should ascertain whether the student has access to a soil source (yard, garden, pond, etc.). If they do not, the instructor could provide curbside pickup of bags of sediment, or perform a remote observation project. If families do not want to keep the column inside, they can be kept outdoors where they can receive sunlight, but they should be stabilized to prevent spills. The instructor could set up several control and light columns, then post photos or short videos for students to observe. Ideally, students will make their own columns and post photos as they progress, allowing classwide interaction and comparison.
Conclusion
The Winogradsky column is an inexpensive and simple phenomenon to use for investigating cellular processes in the classroom or at home. Students learn that photosynthesis requires light and can be performed by microscopic organisms rather than plants alone. They also learn that microbial growth produces waste gases, and they hone their observational skills by recording notes and illustrations of the columns as the microbial communities slowly form over time. •
Acknowledgement
This professional development MWEE project was funded and supported by Western Connecticut State University (WCSU), WCSU’s Finding Our Way project, supported by the National Oceanic and Atmospheric Administration’s, National Marine Fisheries Service, Office of Education, award through its New England B-WET Program # NA16NMF0080003 to TP.
ONLINE Resources
Blog on how to build a giant Winogradsky column—https://schaechter.asmblog.org/schaechter/2018/08/how-to-build-a-giant-winogradsky-column.html
Museum of Natural History directions for classic Winogradsky columns—https://www.amnh.org/explore/ology/microbiology/make-a-home-for-microbes
Popular Science article on growing a bacterial zoo in a bottle—https://www.popsci.com/grow-bacterial-zoo-in-bottle/
Scientific American article on making a Winogradsky column—https://www.scientificamerican.com/article/bring-science-home-soil-column/
Winogradsky column video—https://www.youtube.com/watch?v=Arb-kHfSe4E&feature=youtu.be
Worldwide Winogradsky Project, with directions for teachers—https://sites.udel.edu/winogradsky/
Hannah T. Reynolds (reynoldsh@wcsu.edu) is an assistant professor and Theodora Pinou is a professor, both in the Department of Biological and Environmental Sciences at Western Connecticut State University in Danbury, Connecticut. Joy Pires, Jeffrey Brewster, and Nicole Lalier are science teachers at Broadview Middle School in Danbury, Connecticut.
Biology Disciplinary Core Ideas Inquiry Labs Life Science Phenomena Teacher Preparation


