feature
Biosand Filters for Water Purification
Authentic Science and Engineering
The Science Teacher—March/April 2021 (Volume 88, Issue 4)
By Jawaher Alsultan, Madison Rice, Allan Feldman, Tara Nkrumah, Sarina Ergas, and Kebreab Ghebremichael

Currently more than 800 million people worldwide lack basic drinking water service. In 2017, 435 million people used unimproved drinking water from unprotected wells or springs, and 144 million people collected drinking water from rivers, lakes, or other surface sources of water that may be unsafe (UNICEF 2019). The use of unsafe water as a source for drinking, cooking, and bathing can lead to serious health complications. Each year, nearly 300,000 children under the age of five die due to diarrheal diseases, which is easily preventable through proper drinking water and sanitation infrastructure and education (WHO 2019). Point of Use (POU) water treatment technologies can play an important role in making safe and clean drinking water available to communities.
A number of POU water treatment technologies are used worldwide, such as solar disinfection and boiling. One low-cost, low-technology POU system is the biosand filter (BSF). BSFs offer an effective household-level treatment that is being used in developing countries. BSFs have three layers of media: a coarse gravel underdrain at the bottom, a separation layer of finer gravel, and sand as the main filter medium. A sticky biolayer (also called a “schmutzdecke”) forms naturally at the top of the sand because there is a layer of water above the sand (see Figure 1). Water that needs to be treated is poured (or “charged”) into the BSF once or twice each day. The biolayer traps particles and pathogens, such as the bacteria and viruses that cause human illness. During the “pause period” between charges, the “good” bacteria in the biolayer prey on the trapped pathogens. BSFs can be constructed with locally available materials such as concrete, plastic containers, sand, and gravel (CAWST 2012).
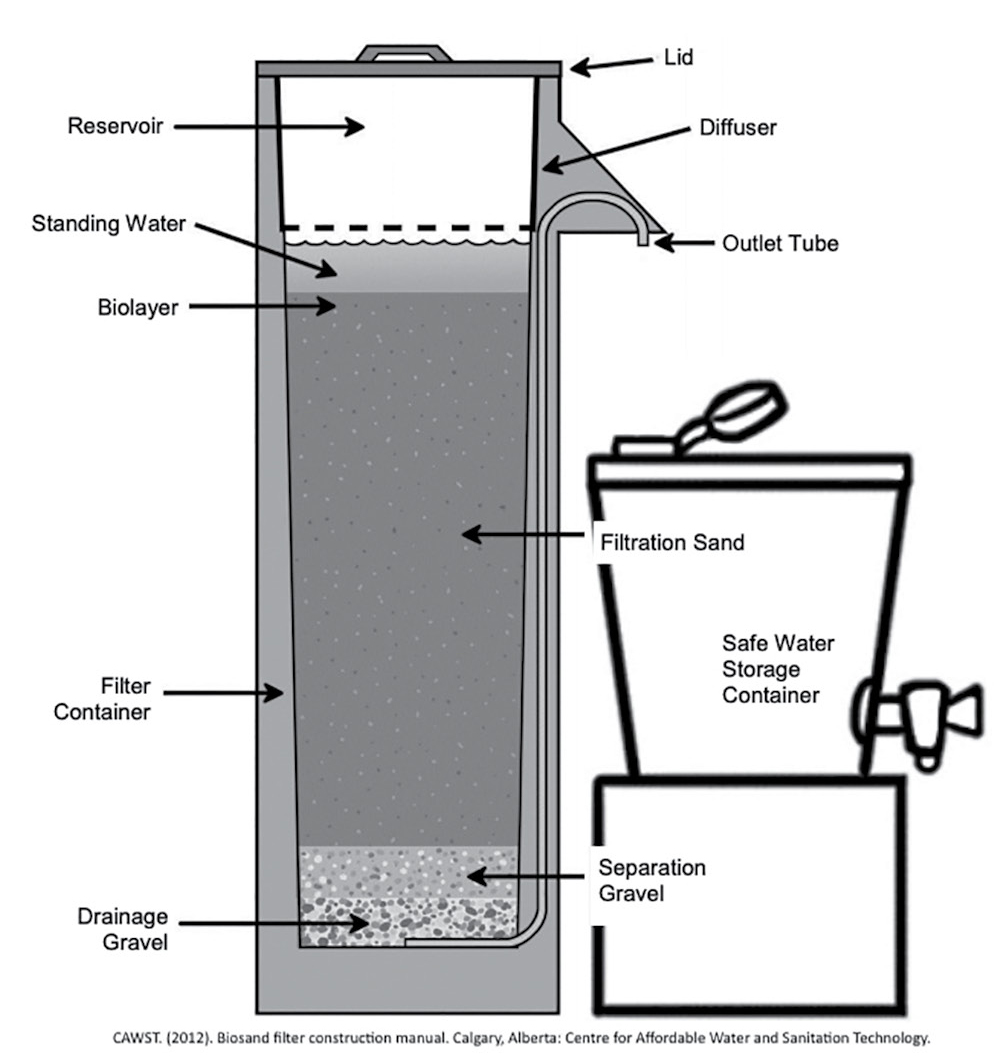
Schematic diagram of the parts of a biosand filter.
This activity is best begun by providing students with background information about the need for POU water treatment, including content such as the amount of fresh water available for human use, why there are shortages in particular places, and how water gets contaminated. A focus on the shortage of potable water and the use of BSFs to respond to this problem opens up a wide range of other science content (disciplinary core ideas) that can be addressed as the students engage in the science and engineering practices (SEPs). Topics such as adsorption, filtration, and biodegradation as the water flows through the BSF; the need for enough hydraulic head to cause the water to move through the BSF; and the biological processes that results in the growth of the biolayer and how beneficial microbes compete with pathogens could all be addressed.
For example, one of the suggested measurements is for the indicator bacteria, E. coli, which provides information about whether there are bacteria in the water that could be harmful to people. These bacteria can cause stomach cramping, diarrhea, or nausea and vomiting. The latter two effects can cause severe dehydration, which is the cause of death for many children who lack potable water. However, because of the interdisciplinary nature of the activity, it can be used as the impetus to explore a wide variety of science content. Therefore, it is up to teachers to determine where this would fit best in their curriculum, which would then determine the particular DCIs that would be addressed.
Engaging in the practices of science and engineering
The Framework for K–12 Science Education (NRC 2012) and the Next Generation Science Standards (NGSS Lead States 2013) call for students to develop and use SEPs. This can be done by engaging students in authentic research and design activities similar to those of scientists and engineers (Lee and Songer 2003; Schwartz and Crawford 2004), and even further, by having the students engage in the practices concurrently with scientists and engineers who are investigating similar questions using similar methods (Chapman and Feldman 2017).
Engineers and scientists at our local university are studying ways to improve the efficacy of BSFs. Beginning in 2018, the team of engineers, scientists, and science educators, along with their graduate and undergraduate students, collaborated with high school teachers in Florida and Ghana to engage students in SEPs using BSFs. The investigations done by the high school students paralleled the studies done at the university. Students built bench-scale models of BSFs that they used to investigate how different variables affected their operation and efficacy to purify water.
The bench-scale BSFs are more than simple physical models to demonstrate filtering techniques. They can be used by the students to engage in authentic SEPs as they manipulate variables to test their hypotheses based on their understanding of how BSFs work (Feldman, Cooke, and Ellsworth 2010). In our work in schools, students asked questions about the characteristics of the BSFs and their operation; planned and carried out investigations to understand how different ways of operating the BSFs affect treated water quality and flow rate; collected and analyzed data about the influent and product water; constructed explanations linking the design and operation of their BSF to water quality; and presented their methods and the arguments that support their findings to their peers and the university team. In addition, they used the results of their investigations to suggest modifications in the design of the BSF as part of the engineering design process.
Asking questions
Before devising their research questions, students first need to learn about the worldwide problem of lack of potable water, and how BSFs can be used to address this problem. They also need to learn about BSFs: how they filter water, and about their components, operation, and limitations. (This information is in a PowerPoint and informational booklet on the project website.) This exposes them to a meaningful science and engineering problem that offers multiple solution paths and multiple solutions (Cunningham and Carlsen 2014).
Students can work in small groups to pose empirical questions about the mechanisms of the BSF and how changes in design and operating conditions could affect its performance. The supplementary materials include a PowerPoint that guides them through the question generation process. Here are some examples of questions asked by students:
- How does the depth of the sand layer affect the quality of the filtered water?
- How does the cleaning frequency affect the quality of the filtered water?
- How does the charging volume affect the quality of the filtered water?
- How does the charge volume affect the filtration rate?
Designing the experiment
Once the students develop the research questions, they need to design their experiment. One student group needs to maintain a BSF as the control. The other student groups need to decide on how they will modify the contents of the BSF column, the charge rate, cleaning frequency, or other variable, and then operate the BSF in a manner that is consistent with their research design.
Building the model BSFs
In building the BSFs we used low-cost or locally found materials as much as possible. Materials can be purchased at local hardware or home improvement stores. Plan to have your students construct a column for each group (see the project website for detailed instructions: http://bit.ly/BSF_TeacherMaterials). It is important that all relevant safety issues need to be addressed in the construction of the BSFs: Students should wear goggles and ear protection and follow safe operating procedures when using tools, such as drills or saws. Students should work with PVC cement in a well-ventilated area and wear latex or nitrile gloves.
The body of each column is constructed from a 4 ft. (120 cm) length of 3 in. diameter PVC pipe (nominal pipe and fitting sizes given) (see Figure 2). A 3 in. PVC elbow and reducing bushing are glued to the bottom of each column. A length of flexible tubing goes from the bottom of the column to a height sufficient to maintain the water level above the sand layer. The tubing is attached to the reducing bushing using a barbed fitting and is strapped to the side of the column. A diffuser plate needs to be placed at the top of column so as not to disturb the sand and the biolayer when charging the BSF. We made the diffuser plate from perforated bottoms of 2 L plastic soda bottles, small bowls, or flowerpots. We constructed simple frames from readily available lumber (Figure 3) to keep the columns upright. The construction of the BSFs and the supporting frame could also be done in a makerspace or technology classroom.
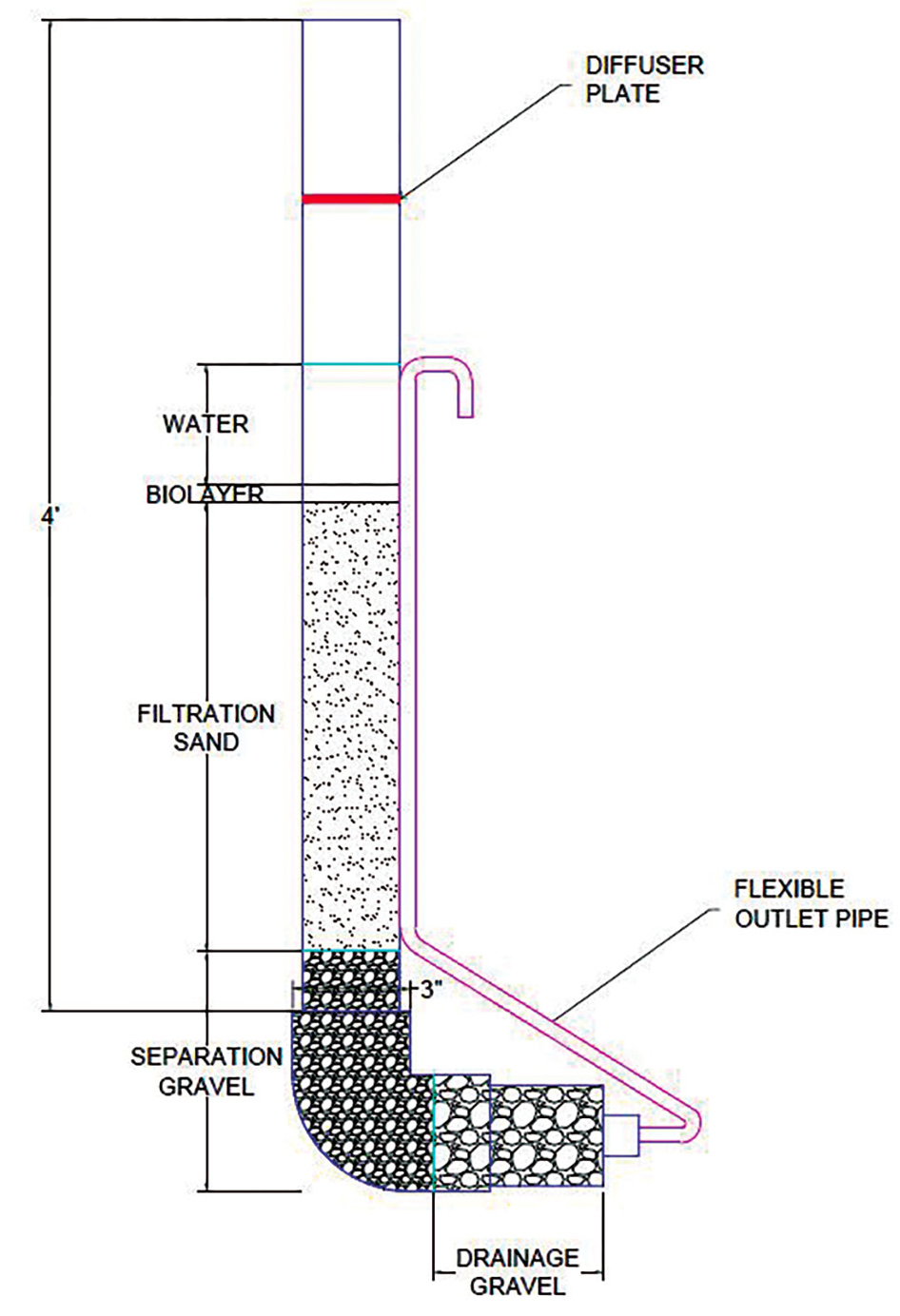
Parts of the BSF columns used by high school students.

One of the participating teachers with the BSFs her students built fixed to a wood frame.
The next step is to fill the BSF columns with gravel and sand. We recommend doing this after the students develop their research questions because their experimental setup could require different depths of sand, or even the addition of another medium such as activated charcoal. The first step is to add water to about half the column depth to prevent the formation of air pockets. Then add 2 in. (5 cm) of coarse gravel (6–12 mm diameter) as the underdrain. Above that is a 2 in. (5 cm) layer of finer separation gravel (0.7–6 mm), and then the top layer—16 in. (40 cm) layer of sand (< 0.7 mm) that serves as the filter medium. The size of sand is very important—if it is too coarse, then you won’t get good treatment of the water, but if it is too fine, the water will flow too slowly. We recommend concrete sand but feel free to improvise (playground sand may work well). The sand must be cleaned very carefully before adding it to the columns to remove fine silt or the filtered water will be turbid.
Operating the model BSFs
The operation of the BSFs is quite simple—each day the BSF is charged with water from a nearby surface water body, such as a stream, canal, or stormwater pond. Students need to be carefully supervised if they collect the water. If the water body does not meet recreational water quality guidelines (i.e., for swimming, wading, or fishing), students should wear goggles, lab gloves, and lab aprons or coats. The typical charge is 1L/day from Monday to Friday. As the BSF is operated, the biolayer will grow and start trapping particles and bacteria. The filtered water quality will improve over time but also the filters will start to clog and the flow rates will decrease. When the BSF becomes so clogged that the flow rate is unacceptable to the user, it is time to clean it using the “swirl and dump” method. A stick can be used to agitate the biolayer and water is scooped out of the top. Be careful not to disturb the sand layer itself. After cleaning, the biosand filter can be operated as usual. You should see the flow rate improve but the water quality might degrade for a few days.
Data collection
To get sufficient data, the BSFs need to be operated for at least four to five weeks. Typical data are pH, turbidity, flow rate, and the presence of harmless fecal indicator bacteria (E. coli). All measurements are made on the influent and the effluent, except for the flow rate. While data collection for most of the parameters should be made on a weekly basis as soon as the students begin operating the BSFs, E. coli measurements should begin after the first 14 days of operation, which is when the biolayer is expected to develop. Detailed instructions for making the measurements as well as homemade instruments are on the project website. Lab safety procedures need to be followed carefully during the data collection process. Goggles, lab coats or aprons, and protective gloves should be worn. Special care should be taken while doing the fecal indicator bacteria tests. Used plates must be disinfected with a 10% bleach solution prior to disposal.
Analyzing and interpreting data, and communicating results
While the data are collected, the students analyze them for their specific BSF, and compare their results against the control. They also can compare them with the other groups’ data. Based on their data, the students argue if and how the modifications they made to the BSF or its operation affected its performance and the quality of the effluent water. They can also suggest modifications to improve the efficacy and ease of operation of the BSFs. Students can support their arguments with graphs and tables of their data and the control. Each student group can then prepare a written report, presentation, or poster, depending on how and with whom the results will be shared (Figure 4 and project website).
Pages from a booklet produced by a group of high school students about their research.
Three-dimensional learning, assessment, and differentiation
The activity described here is truly interdisciplinary and exemplifies the connections among science, technology, engineering, and mathematics. Students engage in science and engineering practices as they develop research questions and hypotheses, collect and analyze data, and present arguments for their findings. In the NGSS table (see Online Resources) we focus on disciplinary core ideas (DCIs) from life science, Earth science, and engineering design. However, other DCIs could be explored, such as HS-PS1 Matter and Its Interactions. Therefore, this article’s standards table should be considered a starting point for teachers as they incorporate this activity into their existing curricula.
An activity such as the one described in this article is best when it is incorporated into the existing science curriculum. As such, there would be assessments that teachers are already using that can uncover what students learn as they engage in the SEPSs. However, some teachers might want to take a closer look and assess the learning of the practices. There are a wide variety of rubrics available on the Internet for assessing the learning of the SEPs (see Online Resources) is a rubric that we developed for BSFs. Another example is the Engineering Design Process Performance Assessment Rubric from the Knowles Teacher Initiative (https://knowlesteachers.org/wp-content/uploads/2017/10/KnowlesEngineering_DesignProcessRubric.pdf). Other assessment resources are available from nextgenscience.org and NSTA.org.
This project can easily be modified for students with varying needs. For example, teachers can carefully scaffold the development of the research questions, and help the students fine tune them to meet their needs. Learning to make measurements can be a group exercise where students record measurements for the BSF on the whiteboard to compare and contrast their results. To accommodate large class sizes, students can be placed into groups of five or six and be assigned rotating roles, such as sample collector, sample analyzer, data recorder, charger, or team leader. If materials are limited, the entire class can test the same variables. Data analysis and the preparation of the final product can also be scaffolded. Of course, teachers should seek out help from special education or ESOL teachers to make more significant modifications.
Conclusions
Hundreds of millions of people worldwide have limited access to safe, clean drinking water. Although for most Americans this problem may seem very far removed from their experience, there are many resources available on the internet that can bring the reality of water scarcity into the classroom (see the project website). We have found this to be a problem that resonates with many students when they become aware of how it affects people their own age.
Experimenting with BSFs is a way for students to participate in solving the problem of water scarcity, poor water quality, and inadequate sanitation that have negatively impacted the health and livelihoods for families around the world. In addition, it can provide students with a voice and empower their capacity in STEM in two ways, first by their authentic engagement in the SEPs, and second, by investigating ways to enhance the efficacy and operation of BSFs that could help those in need of an inexpensive way to purify their water. ■
Acknowledgements
We would like to acknowledge the help of the following University of South Florida students: Michelle Henderson, Xia Yang, Derek Erickson, Itze Kenney, Mercedes Navarro-O’Hara, and Aubrey Selamu-Bell for their help developing the project materials, and working with the participating teachers and students. We also want to acknowledge the teachers from Hillsborough County Public School District who worked with us in the development and implementation of the materials, and invited us into their classrooms. In addition, we want to thank our hosts in Ghana including Dr. Richard Buamah of the Kwame Nkrumah University of Science and Technology, and the teachers, students, and administrator of Kumasi Academy. Finally, this project would not have been possible without the support of the Joy McCann Foundation, and the National Science Foundation under Grant No. OISE-1827132.
Online Resources
Project website: http://bit.ly/BSF_TeacherMaterials
Rubric: https://bit.ly/3tgu6SX
Connecting to the Next Generation Science Standards: https://bit.ly/3iXCstP
Jawaher Alsultan is a doctoral candidate at the University of South Florida, Tampa, FL, Madison Rice is an engineer at Kimley-Horn and Associates, Tampa, FL, Allan Feldman (afeldman@usf.edu) is Professor of Science Education at the University of South Florida, Tampa, FL, Tara Nkrumah is a post-doctoral fellow at Arizona State University, Tempe, AZ, Sarina Ergas is Professor of Environmental Engineering at the University of South Florida, Tampa, FL, and Kebreab Ghebremichael is Associate Professor of Global Sustainably at the University of South Florida, Tampa, FL.
Biology Earth & Space Science Engineering Environmental Science Interdisciplinary Life Science Science and Engineering Practices High School


